
Regulatory
Latest News
The FDA has approved a new single-injection, once-monthly maintenance regimen for Eli Lilly’s Omvoh (mirikizumab-mrkz), offering adults with moderately to severely active ulcerative colitis a more convenient dosing option that maintains proven efficacy and long-term remission outcomes.

FDA Expands Indication for Merck’s Winrevair in Pulmonary Arterial Hypertension

FDA Approves Bayer’s Lynkuet as First Dual Neurokinin Antagonist for Menopausal Hot Flashes
Latest Videos

More News

An analysis of pricing data for 12 high-revenue drugs from Medicare Part B and Part D compares US wholesale acquisition costs with the lowest prices in reference countries under the most-favored nation model.

Amid converging pharma pricing trends, such as those triggered by the Inflation Reduction Act, companies must adopt novel operational practices and strategic approaches in line with new parameters for success across a product’s lifecycle.
Sanofi's Tzield gains FDA's expedited review, promising rapid access to innovative treatment for type 1 diabetes and addressing significant medical needs.

Amgen and AstraZeneca’s Tezspire (tezepelumab-ekko) gains FDA approval as an add-on maintenance therapy for patients aged 12 and older with inadequately controlled chronic rhinosinusitis with nasal polyps.
The President claimed that prices would drop significantly based on government negotiations.

EMD Serono partners with the U.S. government to enhance IVF access, offering significant discounts on fertility treatments for millions of women.

Efforts to regulate pharma prices will impact other sectors of healthcare, with PBMs likely to feel the impact.

At the heart of this moment is a growing unpredictability in how U.S. healthcare policy is being shaped.

The company’s CEO will reportedly joining the President to announce the deal.

Multiple pharma companies have already announced plans to invest in domestic projects.

In the second part of Pharm Exec’s webinar series, experts discuss the changing landscape surrounding the MFN executive order.

Serialization changes what's possible.

Pharma companies have the choice of facing strict tariffs or negotiating with the current administration.

In today’s Pharmaceutical Executive Daily, we cover the FDA lifting its clinical hold on Neurizon Therapeutics’ lead program Nuz-001, the agency’s export ban on Sun Pharma’s Halol facility, and new insights on how small biotechs can better navigate regulatory and market complexities.
CDC introduces shared-decision making for COVID-19 vaccinations, emphasizing informed consent and individual consultation with healthcare providers.

Products tested and manufactured exclusively from domestic materials will be eligible for the program.

This marks the latest change in the President’s strategy.

How cell and gene therapy developers can lessen the chances of receiving FDA complete response letters through the use of proactive chemistry, manufacturing, and controls strategies.
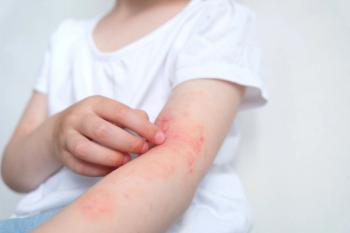
The FDA has expanded Johnson & Johnson’s Tremfya (guselkumab) approval to children six years and older weighing at least 40 kilograms with moderate to severe plaque psoriasis or active psoriatic arthritis, marking the first pediatric approval of an IL-23 inhibitor for these conditions.

Companies that have broken ground or begun construction on US manufacturing sites will be exempt from these tariffs.
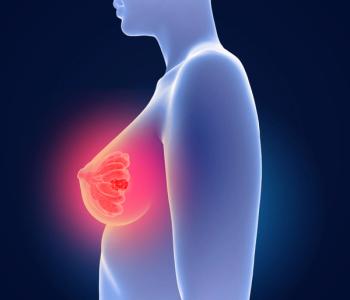
FDA approves Eli Lilly’s Inluriyo (imlunestrant) for adults with ER-positive, HER2-negative, ESR1-mutated advanced or metastatic breast cancer that has progressed after endocrine therapy.

Aside from pricing changes, the impacts that MFN could have on the US market could have global reverberations.

In order to enforce MFN, regulatory agencies must navigate the complex process of determining a drug’s true value.

The President announced FDA will no longer recommend the use of the common painkiller during pregnancy, despite a casual link between the medication and autism not being established.

Alongside regulations from the IRA, oncology R&D is set to face unique challenges when MFN is enforced.











