Capivasertib (Truqap) plus fulvestrant (Faslodex) is a first-of-its-kind combination for HR-positive, HER2-negative locally advanced or metastatic breast cancer with one or more PIK3CA, AKT1 or PTEN biomarker alterations.

Alladapt Immunotherapeutics' Oral Immunotherapy for Multiple Food Allergies Gets FDA Fast Track Designation

Capivasertib (Truqap) plus fulvestrant (Faslodex) is a first-of-its-kind combination for HR-positive, HER2-negative locally advanced or metastatic breast cancer with one or more PIK3CA, AKT1 or PTEN biomarker alterations.

Emerging technologies like digital twins offer supply chain efficiency solutions, allowing the industry to integrate sustainable practices while ensuring patient safety.

Repotrectinib (Augtyro) is a next-generation, potential best-in-class tyrosine kinase inhibitor approved to treat locally advanced or metastatic ROS1-positive non-small cell lung cancer.

Webinar Date/Time: Tue, Dec 12, 2023 10:00 AM EST | 9:00 AM CT | 3:00 PM GMT | 4:00 PM CET

When considering factors like disease biology and patient needs in drug discovery and development, the goal is to deliver effective and scalable treatments globally by balancing established approaches like small molecules or protein therapeutics with more complex modalities such as RNA, cell, and gene therapies.
Defencath is indicated for the prevention and treatment of catheter-related bloodstream infections in adults with kidney failure administered chronic hemodialysis via a central venous catheter.

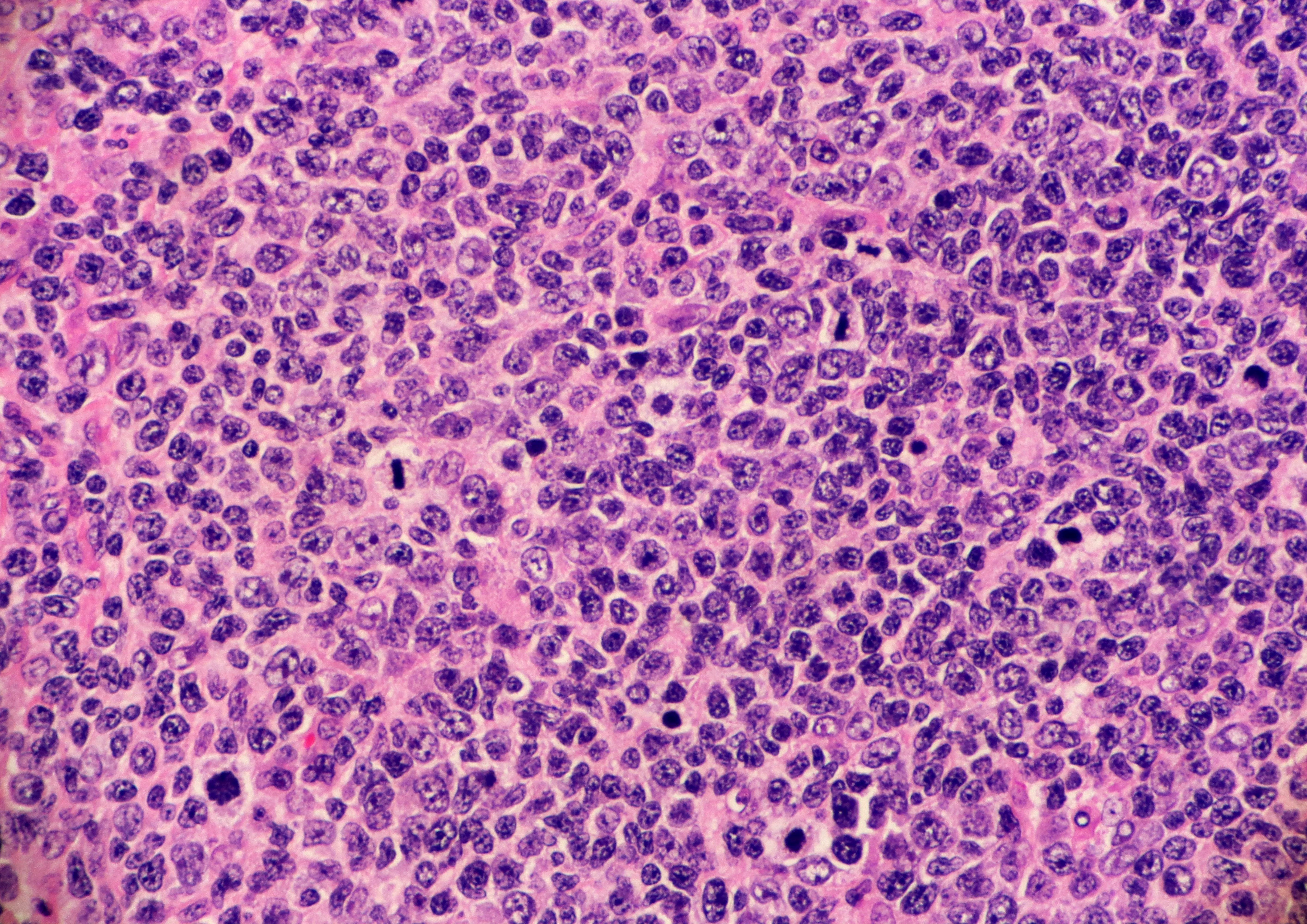
The FDA granted accelerated approval to Aliqopa in September 2017 for the treatment of adults with relapsed follicular lymphoma previously treated with at least two prior systemic therapies.
FDA expands indications for bupivacaine liposome injectable suspension (Exparel; Pacira BioSciences, Inc.) for use in adult patients as an adductor canal block and a sciatic nerve block in the popliteal fossa.

The global market for chikungunya virus vaccines is estimated to exceed $500 million annually by 2032.
Supplemental Biologics License Application for lisocabtagene maraleucel (Breyanzi) seeks to expand the current indication include the treatment of adults with relapsed or refractory chronic lymphocytic leukemia or small lymphocytic lymphoma who were previously treated with a BTKi and BCL2i.
Adalimumab-bwwd (Hadlima; Samsung Bioepis Co., Ltd. and Organon & Co.) is seeking to become the third Humira biosimilar deemed interchangeable with the reference product.
Adzynma is the first approved genetically engineered protein product for the treatment of patients with congenital thrombotic thrombocytopenic purpura.

Solomon discusses new ways that researchers are fighting against bacterial resistance to medications.

A look into the potential effect of psychedelic drugs on various illnesses, such as mental health conditions.
FDA restricts use of pembrolizumab (Keytruda) combination in the treatment of gastric cancer to patients with certain tumor types.

Incidence rates have grown for leptomeningeal metastasis, a rapidly progressing and fatal complication of several cancers.

Ongoing phase 1 dose-escalation trial of KO-2806 (FIT-001) for the treatment of patients with KRASG12C-mutated non-small cell lung cancer expected to begin dosing patients in combination with adagrasib by mid-2024.
The newly approved Alinity m high risk human papillomavirus (HPV) assay is indicated to detect HPV and for use in routine cervical cancer screening per professional medical guidelines.

Phathom Pharmaceuticals announced that it anticipates vonoprazan (Voquenza) to be commercially available by December 2023.

Webinar Date/Time: Thursday, December 7, 2023 at 10am EST | 7am PST | 3pm GMT | 4pm CET
SLS009 is a novel CDK9 inhibitor under investigation for the treatment of relapsed/refractory peripheral T-cell lymphomas.

Exagamglogene autotemcel (exa-cel) has shown the potential to be a landmark therapy in preventing episodes of excruciating pain among patients with sickle cell disease.
The branded form of secukinumab is currently the only FDA-approved fully human biologic that directly inhibits interleukin-17A.

Abatacept is indicated across multiple inflammatory conditions, including for the treatment of adult patients with moderately to severely active rheumatoid arthritis, pediatric patients with moderately to severely active polyarticular juvenile idiopathic arthritis, and active juvenile psoriatic arthritis.

The FDA granted Wezlana with interchangeable designation after clinical trials found no clinically significant differences in safety and efficacy for the indicated conditions across multiple inflammatory diseases.