Breath of Hope: TB in Africa
Pharmaceutical Executive
In The Small, Dusty, District Hospital In Temeke, Tanzania, head nurse Sarah Wilson Ochogo is carrying out her morning ritual. She drags a table outside the hospital's tuberculosis (TB) unit, sets out big, white, plastic bottles of pills, and opens a pencil-lined ledger that tracks her patients, their drugs, and their doses. By seven, more than 100 patients are lining up. They place their green patient cards under a stone near the table, and sit down on wooden benches to wait until Ochogo calls their name, gives them their medication, and watches while they take it. By a bit after nine, the patients are off to their daily routines-drawing water, tending crops, or just getting by the way people always have in Africa. And all of them, with luck, are one more day closer to curing their TB.
IN THE SMALL, DUSTY, DISTRICT HOSPITAL IN TEMEKE, TANZANIA, head nurse Sarah Wilson Ochogo is carrying out her morning ritual. She drags a table outside the hospital's tuberculosis (TB) unit, sets out big, white, plastic bottles of pills, and opens a pencil-lined ledger that tracks her patients, their drugs, and their doses. By seven, more than 100 patients are lining up. They place their green patient cards under a stone near the table, and sit down on wooden benches to wait until Ochogo calls their name, gives them their medication, and watches while they take it. By a bit after nine, the patients are off to their daily routines—drawing water, tending crops, or just getting by the way people always have in Africa. And all of them, with luck, are one more day closer to curing their TB.
TB Incidence Rate
Throughout Africa—indeed, throughout the world—the same ritual is taking place, as healthcare workers deliver TB drugs to millions of patients spanning countries, languages, and cultures. It's called DOTS—"directly observed short-course treatment"—and it is the internationally recommended strategy for TB control, put forth by the World Health Organization (WHO). Patients in DOTS take a two-month daily course of the antibiotics rifampacin, isoniazid, pyrazinamide, and ethambutol, followed by either four months of rifampicin and isoniazid, or six months of isoniazid and ethambutol.
Certainly, the DOTS strategy aims high: to ensure that patients take their drugs in the same, correct way and for the full course of treatment. That's partly to ensure that they recover—but partly to protect against half-completed treatment that gives rise to mutated, drug-resistant strains of the disease, which are more difficult and expensive to treat.
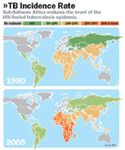
When we think of TB epidemics, we tend to think of the past, especially the great epidemic that swept Europe in the 19th century. But in fact, TB is one of the world's great killers again: Two million people die each year from the disease. WHO estimates that more than two billion people worldwide, or one third of the world's population, carry at least a latent TB infection. And the AIDS epidemic is making it worse. TB accounts for a third of AIDS deaths worldwide; the co-infection is commonly referred to as the "deadly duo." In 2004, an estimated 14 million people were living with dual HIV and TB infections. Seventy percent of them were African.

From AIDS to TB
Drugs are available and cheap—a six- or eight-month course of treatment procured through generic Indian manufacturers costs about $10. The guidelines for DOTS administration are well known. The supply-chain management for the procurement and administration of the drugs is in place, with tight controls to prevent diversion. There's just one problem: It isn't working. Millions have died and millions more are dying. And in the past few years, pharma companies have discovered that there is a real role for them to play in fighting TB. Several factors play into that realization:
- Globalization is transporting diseases from one part of the world to others. That has already happened with TB in New York City, where infected immigrants are thought to have contributed to the resurgence of the disease in the late 1980s.
- TB is a death accelerator of AIDS patients, making it a global health priority, and a target of the United Nation's Millennium Development Goals. The public and other stakeholders will likely exert pressure on the industry to contribute its technical and public-health expertise.
- The growing number of bacteria resistant to today's TB therapies has given pharma a clear-cut role of bringing its unique skills in drug discovery to the table.
- Academics and nonprofits are settling into a new way of partnering with industy that allows them less costly and risky ways to participate in the search for new TB drugs.
- Governments and other third parties will increasingly look to pharma's infrastructure initiatives, many of which were established to fight AIDS, to help manage the TB burden.
TB advocates complain that there have been no new TB drugs for 40 years. Certainly, pharma never had it easy in TB—and it won't get better soon. New science must harvest innovative therapies; new partnerships must develop treatment for patients and entire countries without the resources to pay market prices. And new programs must deliver those treatments on the ground.
The Search for Basic Science
The first anti-TB drug, streptomycin, was discovered in 1944 by Selman Waksman at Rutgers University—funded by a grant from Merck—which made the disease treatable for the first time in its long history. Following streptomycin, other TB agents were introduced, including isoniazid (1952), pyrazinamide (1954), ethambutol (1962), and rifampicin (1963). Researchers soon demonstrated that TB therapy was more efficient, and less likely to end in drug resistance, when they switched from single-drug regimens to combination therapies using multiple drugs.
In 1969, following the discovery of these and other antibacterial drugs, US Surgeon General William Stewart proclaimed it was "time to close the book on infectious diseases." And indeed, the book closed—though not quite in the way Stewart meant. The disease continued largely unabated outside the United States, and the evolution of treatment stood still.

Corporate Conscience
The result is that today's therapies are good—they work—but they have their problems. "Rifampacin is the best of drugs and the worst of drugs," says Balganesh Tanjore, head of research at AstraZeneca's TB-focused site in Bangalore, India. He says that rifampacin is powerful against the disease, but the toxicity of the drug can outweigh the benefits.
Add poverty to the mix, and the margin for error grows tighter: "Most of the people are very poor," says Ali Hamisi, MD, from Tanzania's Bagamoyo District Hospital, where TB is a top killer. "If they are not well-fed, toxicity is very high." What's more, rifampacin is contraindicated with several AIDS therapies, and pyrazinamide is unsafe for pregnant women.
The biggest challenge of TB treatment, however, is maintaining compliance during the long course of therapy. "Why can't TB have a standard 10-day treatment, just like any ordinary antibiotic?" asks Marie Freire, who heads the Global Alliance for TB Drug Development, an organization working to increase industry participation in TB R&D. According to WHO's recently published "Global Plan to Fight TB, 2006-2015," that's unlikely to happen before 2050. The basic science of the disease is not yet there.
"When you talk to various communities, you realize the understanding of how a TB bacillus behaves in humans is virtually non-existent," says Ken Duncan, consultant to the Bill and Melinda Gates Foundation, and former GlaxoSmithKline executive.
TB researchers also lack some important research tools, such as targets, biomarkers, and predictive models to tell scientists if what works in cell cultures will work in man. "Most drugs so far are developed against TB in test tubes," says Douglas Young, a professor of medical microbiology at the Imperial College London, and a recipient of a Gates Grand Challenge grant to develop new treatments for latent TB infection. "We want to choose a defined target—a protein or enzyme that if inhibited, has a good chance of killing the disease—based on an understanding of the physiology of the bacteria as it exists inside our body. If we choose targets that way, then the drugs will be more effective."
The tuberculosis genome has been known since 1988, but so far that hasn't translated into a library of targets. And researchers have only recently begun to look for biomarkers or other key diagnostics. "In the case of HIV/AIDS, you have viral load as a surrogate marker, or CD4 counts as a biomarker," says Freire. "We don't have that. We have to wait 18 months for people not to get sick [to know if the drug works]."
As these tools come together, it becomes far more practical for the industry to be involved in TB research. "The pharma industry can come in and bring along that knowledge and experience of what targets are potentially 'druggable,'" says Duncan. "Then we have a realistic potential of actually making a drug, rather than just doing academic research."
A New Framework for Research
The world needs new, more powerful TB drugs. In the short run, that probably means repurposing existing compounds. For example, studies show the fluoroquinolones gatifloxacin (better know as Tequin), and moxifloxacin, (the brand Avelox), can help reduce TB treatment to four months from the current six or eight months when substituted for ethambutol.
But in the long run, the real progress should come as new drugs emerge from the pipeline. That may not be too far off, given the way companies are more actively participating in developing drugs for neglected diseases. Before 2000, companies that conducted R&D in this area largely did so alone, forcing them to carry out large, late-stage clinical trials in the developing world. However, the cost and the risk of these types of ventures made it unsustainable.
In the last five years, however, pharma has developed new ways of partnering and has shifted its focus to discovery and early-stage research. Today, it is far more likely that a company working in neglected diseases will hand off the late-stage R&D to a non-profit or NGO partner. This strategy is working: While only 13 new neglected-disease drugs were introduced from 1975 to 1999, there are now 63 active projects in development, which should yield eight or nine new therapies by 2010, according to a report by Wellcome Trust, an independent UK-based charity funding biomedical research. A third of these active projects are being conducted by three Big Pharma companies: AstraZeneca, GlaxoSmithKline, and Novartis.
"Mind you, this is a double-edged sword, because they don't want to carry the cost of clinical trials," says Freire. "But I'm very excited because at last we have a discovery pipeline."
These companies have taken different approaches to setting up their TB units. Novartis, for instance, has created the Novartis Institute for Tropical Diseases (NITD), a public/private partnership between the company and the government of Singapore. GlaxoSmithKline, on the other hand, has teamed up with the TB Alliance to create a shared research program out of GSK's Tres Cantos site in Spain. And AstraZeneca converted its Bangalore-based R&D site into a center of excellence for TB research in 2001. Novartis pays for its own research; GSK leans on its partner for half of its employees; while AstraZeneca has kept the unit within its ordinary drug-development structure and, some say, sees it as a basis for a potential commercial operation.
Despite their differences, these operations benefit from their relationships with the companies that started them, and they are positioned to also partner with outside organizations.
Commercial libraries Perhaps the greatest advantage pharma's neglected-disease research groups have is access to the commercial libraries of their companies. "That's the starting point for finding new leads," says Paul Herrling, Novartis' head of corporate research and chairman of NITD "We can't give general access to our libraries because that's also the basis of our commercial success. Instead, NITD can screen the entire Novartis library, and just pick out those compounds that work—and make them available at no profit."
Existing infrastructure These groups can also draw on existing infrastructure at the companies for experience, tools, and manpower. For example, in addition to pursuing its own drug discovery, the AstraZeneca site in Bangalore can call on the company's genomics and infection-research operations in Boston, among other sites.
Partner-friendly TB research units must also collaborate externally, not least because partnerships with nonprofits can attract funds, which may be pharma's only chance to offset its TB research investment. It may seem counterintuitive for big companies to turn to nonprofits for funding, but it can make a difference. "You would think that the small amount of money we give Big Pharma is really insignificant," says Freire. "It turns out it is fairly significant because they operate on the model of no profit, but also no loss."
Indeed, today's major funders structure grants in a way that encourages partnerships. "Major funding streams have helped enormously to focus and to bring home the message that we either do it together, or we shall be stuck as we have been over the last three decades," says Alex Matter, MD, who is credited with discovering Gleevec (imatinib) and is now director of NITD.
But despite companies' willingness to partner, says AZ's Tanjore, collaborations between Big Pharma and third parties are still not easy.
For instance, Imperial College's Young, who is working with NITD among other researchers, formally received the Gates Grand Challenge funding on July 1, 2005. "We're just getting into the way it all has to be done under proper legal and confidentiality agreements, because we're working with pharma which eventually will have the intellectual property to protect these things in the end," says Young. "We're finding out how to do it."
The Social Translation
The oldest hospital in East Africa stands in the town of Bagamoyo, which used to be a major port in the slave trade. Now an up-and-coming town, and in recent years, hosted medical meetings on AIDS and tuberculosis—including a major international meeting held there this past October by NITD. But old Africa is still alive and well in Bagamoyo. TB is the leading cause of death in the town, says Hamisi, the hospital's TB coordinator. But even though free drugs are available to treat it, many patients still turn to what's familiar, available, and accessible—the local witch doctors and traditional healers.
In Africa, an epidemic is only half medical. The other half is social. That is especially true with TB. Bagomoyo is a rural district full of poor people, so TB diagnosis and treatment are difficult. The hospital serves 82 villages stretched across 9,000 square kilometers. The majority of people live on less than $1 a day, and often can't afford the time or the cost for transport to the hospital. Sure, clinical officers go into the field, and field sites conduct TB testing. But, with an annual operating budget of $70,000, the hospital can't hope to meet the growing need for treatment.
Certainly, these obstacles won't be overcome with a new drug or pill combination. Instead, pharma must get involved in fashioning effective treatment programs if they want the advances they make in the lab to affect TB care on the ground.
Integration, not separation Before TB drugs, isolation was the only means of controlling the spread of infection (a practice that continues to this day for many patients with multi-drug resistant TB). As such, many TB treatment sites are separate from other health services. The arrangement can make patients unwilling to seek treatment. "When you say, 'All AIDS patients go through this door,' it creates stigma," says Pascience Kibatala, MD, chief medical officer of St. Francis District Hospital, in Ifakara, Tanzania. "The same goes for TB."
A recent renovation of Muhimbili National Hospital, in Tanzania's capital city Dar es Salaam, show that TB care is being moved into outpatient treatment services. (See "From AIDS to TB") And new funds by the Global Fund to Treat AIDS, Malaria, and TB will help to further integrate care by funding joint HIV/TB programs, says Saidi Egwawa, national TB and leprosy program manager at the Tanzania Ministry of Health. That's important because it enables clinicians to better monitor co-infected patients who may experience interactions between rifampacin, a TB treatment, and nevirapine—an AIDS drug popular in resource-poor settings because it is donated by Boehringer Ingelheim to pregnant women and their newborn children. Other companies make it available as a generic.
Invest in infrastructure Like AIDS, TB is so generalized throughout the population that companies can improve the care of patients by investing in the overarching healthcare infrastructure—and in the process, help strengthen the entire system.
That type of thinking is replacing the disease focus that characterized early AIDS programs in Africa. "PEPFAR [The President's Emergency Plan for AIDS Relief] has gone into funding the best labs, the best doctors—the best of everything, really—into centers that have no guaranteed perpetuity after the five-year grant," says Christian Lengeler, a scientist with the Swiss Tropical Institute. The result: Well-funded AIDS programs outside the government-run system have siphoned health workers away from hospitals that were already collapsing under the weight of the epidemic.
New company programs must restore, rather than divide, in-country healthcare services. For example, Sanofi-Aventis—a major producer of rifampacin—is building nine DOTS training centers in South Africa (one per province), in partnership with the Nelson Mandela Foundation and the government, says Robert Sebbag, MD, vice president of the company's access-to-medicine program. The program aims to train 50,000 DOTS workers by 2008, and serves as a pilot initiative for future initiatives.
Community-based programs The TB epidemic continues to grow at the rate of one new infection every second. Public-health experts fear that efforts to scale up treatment will be thwarted by the lack of skilled health professionals, given the daily monitoring required by DOTS is so resource intensive. Therefore, companies interested in creating treatment programs should investigate alternative ways of administering drugs, such as self-administered therapy, particularly during the continuation (two-drug) phase of therapy.
"What happens five kilometers beyond the tarmac road?" asks Klaus Leisinger, president and CEO of the Novartis Foundation for Sustainable Development. "You can go to the last village and find Coca-Cola and plastic combs. As long as this channel is there, how can we use it for pharma? Instead of bringing patients to drugs, we need to think about ways to bring drugs to patients."
A Community DOTS program, which brings compliance measures closer to patients' homes, is being tested in Temeke. Still, even that type of TB control requires personnel, and personnel have to be paid. Traditionally, funders would rather put their money into drugs or infrastructure than salaries, but there are signs that some are rethinking that position. The Global Fund, for example, has thus far dispersed funds only for technical components of TB programs, but Egwawa says it may try to increase the effectiveness of its grants by also underwriting human resources.
Strong alliances Pharma has already learned some hard lessons when it comes to the developing world. "Pharma thinks the right to health is aspirational," says Leisinger. "But wake up, no one else in the world thinks that."
Consider pricing, for instance. "There will be pricing pressure whenever drugs are introduced for neglected diseases," says Sebbag. "We changed the pricing with AIDS, now we are doing the same with TB, and then with malaria."
The recent agreement between Bayer and the TB Alliance offers a remarkable example of the industry embracing the spirit of wider access. Bayer has turned over its $500-million-a-year antibiotic moxifloxacin to the organization to test in clinical trials. If proven effective, Bayer will offer "expansive access" to the drug, despite the risk of the deal undercutting profits on the commercial side. Sources even say Bayer will allow the TB Alliance to check its books to ensure they are offering it at no profit. Says Freire: "I don't know of any other pharma company that has said, 'Yeah, we trust our drug, and yes, we are making a commitment to public health, and yes, we're going to make it affordable. And okay, maybe there's a risk of drugs coming back out [through diversion], but we don't believe so. So we're going to do it because it's the right thing to do.'"
DOTS-Plus: The Next Line of Defense
When it comes to multi-drug-resistant tuberculosis (MDR-TB), people are seeking second-line treatments where first-line defenses left off more than half a century ago. Streptomycin, the first TB drug, has become a second-line drug of desperation.
It's a first-line drug, but not often used because it must be given by injections, says Fabienne Jouberton, procurement officer for WHO's Stop TB department. But it's often used that way in Temeke. Given the scarce alternatives, local rules apply. "Once you have resistance, you're going to use everything you can to cure the patient," says Young.
A patient waiting for his streptomycin treatment pulls down his pants, and slaps his right side, where he wants to receive today's shot. The injection is not only a strain on the resources at the clinic, it's an added expense: Although the TB drugs are free, the syringe is not, and he struggles to pay about 50 cents each day for a new one.
At the moment, most of the world's known cases of MDR-TB are in China, India, and Russia, where drugs were available early on and where compliance was a widespread problem. WHO believes that an MDR-TB epidemic won't be a problem in the near future for Africa, given the late introduction of rifampicin. But epidemiologists are debating over the transmissibility of certain drug-resistant strains. "The information we have for Africa is still very patchy," says Ernesto Jaramillo, a medical officer in WHO's TB/HIV drug resistance division.
In fact, some researchers are concluding that MDR-TB may be more advanced in Africa than previously thought. "In Africa, there's not a lot of surveillance for drug resistance because it requires drug sensitivity testing—which is not included in routine TB care," says Megan Murray, assistant professor of epidemiology at the Harvard School of Public Health, "With our surveillance study, we found outbreaks of highly drug-resistant strains that seem very infectious. Our predictions from mathematical models are that those mutations will proliferate and eventually dominate. It will take a while—maybe even decades—but if we don't focus on MDR-TB, we're going to see a rise, and even a replacement of, drug-sensitive TB."
In other parts of the world that have more advanced drug resistance, clinicians have begun using newer second-line treatments, including capreomycin and cycloserine. At one time, such therapies were quite expensive, costing up to $33,000 per patient, according to the CDC Foundation. They also have significant compliance challenges: MDR-TB drugs are more toxic and require 18- to 24-month treatments.
Given the resources needed to ensure patient safety and compliance, administration of these new second-line drugs was erratic, says Jaramillo. In response, WHO created the Green Light Committee (GLC), a subgroup of the Working Group on DOTS-Plus for MDR-TB, to reduce the price, and promote rational use of MDR-TB drugs. GLC obtains price concessions on drugs, but to take advantage of them, organizations must apply to GLC for a technical review to ensure their project makes rational, thoughtful use of second-line therapies. Based on the numbers, GLC has largely done its job: Since 2000, it has reduced the cost of second-line treatments to $1 a dose and now supplies drugs to 37 projects, says Jaramillo.
WHO's "Global Plan to Stop TB, 2000-05" offered a map of pilot projects to demonstrate that it was possible to treat MDR-TB in resource-limited settings. The second Global Plan offers scale-up plans: Only 10,000 MDR-TB patients were treated in the last five years, the second plan aims to treat almost 900,000.
That's a huge increase, and supply is already problematic for second-line therapies. Since 2003, Eli Lilly has sought to increase production of capreomycin and cycloserine by expanding its facilities and signing technology transfer agreements with companies in India, South Africa, and China. But even when completed, these initiatives are unlikely to solve the problem of very long lead times.
"Companies don't want to take the chance of producing expensive therapies that might not be consumed," says Kathryn Kempton, director of procurement at Partners in Health, an NGO focused on TB. "They often don't start production until we place an order—creating 8-, 12-, or even 24-week lead times—which is challenging for projects. If WHO or others could invest in an existing supply or create more transparency about how many patients there are in the market, that would help increase the production, and make our lives a lot easier."
WHO is investigating the feasibility of maintaining a buffer stock of MDR-TB drugs, which would allow programs to ramp up faster and reduce waiting lists, says Jaramillo. But the world is still facing a dearth of manufacturers that can produce MDR-TB drugs that meet the high standards of WHO's prequalification list.
Looking Forward
After years of neglect, TB is finally starting to receive the attention it deserves. "This latest round of the Global Fund, round five, saw TB get its most significant approvals to date," says Robert Matiru, manager of WHO's Global Drug Facility. "That was a dramatic increase in dollars earmarked for TB compared with the previous four rounds of funds combined."
Novartis also just donated 500,000 TB treatments in fixed-dose combination (FDC) for use over the next five years in Sri Lanka and Tanzania. These FDCs reduce the pill burden from eight to 11 pills per day to three to four. "That's the first collaborative arrangement of that kind that we have established," says Matiru. "In the future, we want more of those agreements with companies."
Better science is also on the way: Young noted that his multi-national research team for the Gates Grand Challenge was zeroing in on five targets that may be instrumental in latent infection and shortening TB treatment times, while NIH scientists have discovered the target acted upon by PA-824, a drug candidate purchased by the TB Alliance from Chiron. Meanwhile, OPC-67683, a compound owned by Otsuka, was found to be "far more potent than any of the existing drugs that we used for comparison," says Vince Lawlor, from the company's TB Drug Project.
More companies have entered the TB research arena, including Sanofi-Aventis and Johnson & Johnson. However, the TB Alliance says it is still not getting the kind of critical participation from other players, like biotechs, which can offer important knowledge in terms of biomarkers and surrogate markers.
New TB drugs haven't come to the market for 40 years, and perhaps the greatest challenge is the lack of TB-specific regulatory guidelines for drug development, including fast-track approval guidelines. The TB Alliance is working to bring that issue to the forefront. It hosted a meeting in December that included FDA and addressed issues such as the design of clinical programs for novel combinations of therapies.
"Trials that change one drug for another take four to six years for the first trial to be approved," says Freire. "Then you drop the second drug, which takes another six years. By the time you replace the four drugs, you're into 24, 30 years. We can't do that, but the question remains: How do you conduct very solid, ethical, and well-controlled trials that have different arms in which you substitute more than one drug at a time?"
As industry begins to iron out these issues, another challenge looms: the gap between registration and adoption of policy, says Tom Kanyok, PharmD, manager of product development for WHO's tropical diseases program. For example, most patients in Tanzania take the eight-month DOTS regimen, but studies show the shorter, six-month regimen (with the continuation therapy of rifampicin and isoniazid) was more effective. However, many countries are still "using up the drugs they have" before moving to the six-month regimen, according to one government source.
These issues seem like a lot for industry to sort out, but companies do gain in return. "If you deal proactively with issues, you have lower reputational, legal, and financial risks," says Leisinger. "I want 85,000 ambassadors working for Novartis going out and saying 'Hey, we are doing great things, I'm so proud of it!' We will have less fluctuations, and more motivated people."
Novartis' Matter says other lessons will be more direct. "Neglected-disease research allows us to experiment with new models of discovery. We learn how to do things with less money. If we find ways to decrease the attrition, we will have learned something that's valuable for the whole industry."
Meanwhile, Nurse Ochogo is learning her own lessons. One of her patients, a young man waiting his turn at Temeke Hospital, is back. He completed a few months of DOTS a while ago. But when he started to feel better, he stopped coming. Soon he grew sick, lost weight. Next, the incessant cough started, accompanied by the bloody sputum. Now he's back in treatment, but his old drugs don't work anymore.
Nurse Ochogo has seen it before. She needs time for these returning patients and is speeding up her morning DOTS reveille. She used to see patients individually. Today, she calls them three at a time.

FDA Outlines Updated Requirement for Placebo-Controlled Trials in Vaccine Research
May 21st 2025In an article recently published by The New England Journal of Medicine, FDA higher-ups Vinay Prasad, MD, MPH; and Martin A. Makary, MD, MPH, wrote that any new COVID-19 vaccine must now be evaluated in placebo-controlled studies.
Addressing Disparities in Psoriasis Trials: Takeda's Strategies for Inclusivity in Clinical Research
April 14th 2025LaShell Robinson, Head of Global Feasibility and Trial Equity at Takeda, speaks about the company's strategies to engage patients in underrepresented populations in its phase III psoriasis trials.
Undertreated but High-Risk: The Hidden Health Crisis Facing 50+ Americans
May 15th 2025Leandro Boer, MD, PhD, VP, US general medicines, Amgen, explains why many high-risk Americans aged 50+ remain undertreated for cardiovascular disease, citing insurance gaps, risk misperceptions, and complex guidelines.
Key Findings of the NIAGARA and HIMALAYA Trials
November 8th 2024In this episode of the Pharmaceutical Executive podcast, Shubh Goel, head of immuno-oncology, gastrointestinal tumors, US oncology business unit, AstraZeneca, discusses the findings of the NIAGARA trial in bladder cancer and the significance of the five-year overall survival data from the HIMALAYA trial, particularly the long-term efficacy of the STRIDE regimen for unresectable liver cancer.
Novo Nordisk’s Sogroya Demonstrates Efficacy in Children with Growth Disorders
May 14th 2025Results from the Phase III REAL8 basket study found that once-weekly Sogroya was effective in children with growth disorders, including those born small for gestational age, with Noonan syndrome, or idiopathic short stature.