
FDA approved Baxfendy for adults with uncontrolled hypertension, introducing a new drug class in a large and competitive market.

FDA approved Baxfendy for adults with uncontrolled hypertension, introducing a new drug class in a large and competitive market.

FDA expanded Enhertu (trastuzumab deruxtecan) use in early HER2-positive breast cancer based on phase 3 neoadjuvant and adjuvant trial data.
FDA granted Compass Pathways with a rolling review for COMP360, a proprietary formulation of synthetic psilocybin, in treatment-resistant depression.

Early clinical data from the CHORD trial supported accelerated approval of the first gene therapy targeting OTOF-related hearing loss under the FDA Commissioner’s National Priority Voucher program.

Advice for making your story pitch stand out in this treatment space—where clearly conveying the "why it matters" is essential.

Tips for turning transactional outreach into a trusted working relationship.

Pharm Exec’s latest sampling of notable biopharma brand approvals and launches all have unique stories to tell—from the winding journeys to innovative “firsts” to new momentum for milestones to come.

Leaders in areas such as drug discovery, clinical development and market access will meet to discuss AI, failing life expectancy, and a new generation of talent.

Elliott Yoo, Senior Media Strategist at epocrates, takes a closer look at the trends in CME consumption and what the future of medical education will be – from the priorities for physicians as they juggle learning with patient care, to what type of activities resonate most with prescribers.

The Maryland facility will create cell therapies for use in future cancer trials.
The $1.3 billion acquisition—which included two aqua manufacturing plants—expands Merck’s portfolio in the veterinary pharma space.
Study finds that individuals who took GLP-1 agonists for a long duration had a lower risk of going on to develop more severe forms of liver disease, including cirrhosis and liver cancer.

The FDA previously approved Dupixent in May 2022 to treat eosinophilic esophagitis in patients aged 12 years and older.

The expanded indication for Zynrelef now includes soft tissue and orthopedic surgical procedures.
Obecabtagene autoleucel is chimeric antigen receptor T-cell therapy under evaluation for the treatment of relapsed or refractory B-cell acute lymphoblastic leukemia.
The Osteoboost device has been shown to slow the loss of bone strength and density in postmenopausal women with osteopenia.
Today's FDA approval amends a previously granted accelerated approval for Balversa (erdafitinib) to treat patients with metastatic urothelial carcinoma whose tumors harbor FGFR3 or FGFR2 alterations following prior platinum-based chemotherapy.
Novocure’s tumor-treating fields plus standard-of-care therapies is under evaluation in patients with non-small cell lung cancer following disease progression on or after platinum-based treatment.

HyQvia is the only subcutaneous immune globulin infusion that can be administered once per month.
Vertex Pharmaceuticals' and CRISPR Therapeutics’ Cas9 therapy Casgevy approved as a one-time treatment for transfusion-dependent beta thalassemia on the heels of its approval last month for sickle cell disease.

Ginkgo Bioworks announces projected total revenue of $250–$260 million for 2023 for large scale data generation and artificial intelligence for biopharma R&D.
RC88 is under evaluation for the treatment of patients with platinum-resistant recurrent epithelial ovarian, fallopian tube, and primary peritoneal cancer.
The BioThrax vaccine is indicated for active immunization to prevent anthrax disease caused by Bacillus anthracis in individuals aged 18 through 65 years.
Axon Therapy uses magnetic peripheral nerve stimulation to deliver a non-invasive treatment for chronic pain related to nerve damage caused by diabetes.
SELLAS Life Sciences Group, Inc’s CDK9 inhibitor SLS009 is being evaluated in an ongoing Phase I/II study in combination with Venclexta and Vidaza for patients with relapsed or refractory acute myeloid leukemia.

SH-105 eliminates the need for powder reconstitution, which Shorla stated will bolster the novel product’s efficiency and lower the risks associated with drug preparation.

Moderna’s vaccine revenue dropped from $18.4 billion in 2022 to $6.7 billion in 2023, however the company anticipates profitability this year powered by COVID-19 vaccine sales and the launch of its mRNA-based respiratory syncytial virus vaccine.
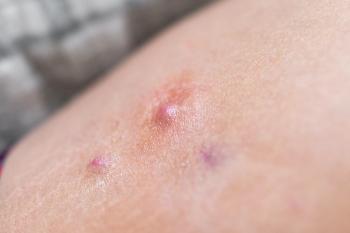
Lutikizumab (ABT-981) will advance to a Phase III trial in patients with moderate to severe hidradenitis suppurativa who previously failed anti-tumor necrosis factor therapy.

Merck has acquired Harpoon Therapeutics with its clinical stage T-cell engager, whereas Johnson & Johnson acquired Ambrx Biopharma with its expertise in next generation antibody drug conjugates and targeted oncologic therapies.

The FDA was unable to approve the Biologics License Application for zolbetuximab because of deficiencies found in a third-party manufacturing facility for the drug, which is under evaluation to treat patients with locally advanced unresectable or metastatic HER2-negative gastric or gastroesophageal junction adenocarcinoma.

March 7th 2022

February 22nd 2023

October 5th 2021

October 19th 2022