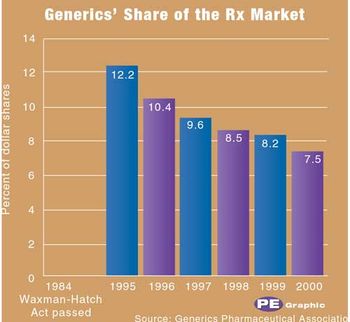
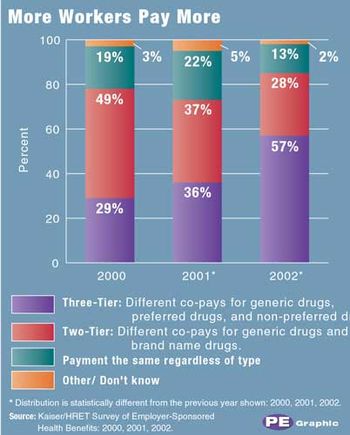
Pharmaceutical Executive
Companies, like individuals, must stay in shape, and both must search-in an environment of high demands on time and resources-for the right tools to achieve and maintain fitness. The pharma industry's current challenges suggest that the need for fitness may be greater than ever: