
Pharmaceutical Executive
One of EMA’s key objectives this year is improvements in dealing with the “causes and impact of shortages of human medicines caused by GMP non-compliance and quality defects”.

Pharmaceutical Executive
One of EMA’s key objectives this year is improvements in dealing with the “causes and impact of shortages of human medicines caused by GMP non-compliance and quality defects”.

Pharmaceutical Executive
A European bid to impose additional limits on research involving human embryos has been defeated.

Pharmaceutical Executive
I’ve been lucky enough to get a sneak preview of Across Health’s 2014 Multichannel Barometer report.

Pharmaceutical Executive
The Obama administration received a substantial amount of good news last month.

Pharmaceutical Executive
Vietnam’s pharma market in Vietnam is set to increase by $5 billion over the next six years.

Pharmaceutical Executive
Turkey is remains a key destination for foreign investment, and the next five years should see renewed interest from pharma multinationals, says a new report by CPhI Worldwide.

Pharmaceutical Executive
Effective clinical trial management depends on accurate and unbiased performance measurement.

Pharmaceutical Executive
FDA, in partnership with other federal and international agencies, has taken action against websites that sell potentially dangerous, unapproved prescription drugs to US consumers.
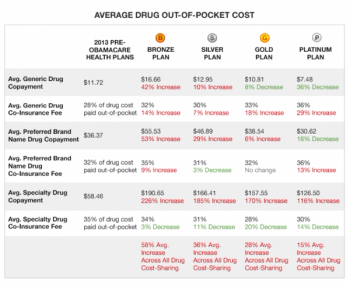
Pharmaceutical Executive
We are now approaching the six month point in the implementation of the new Obamacare program. It’s senseless to go into the good, bad and ugly of this new program.

Pharmaceutical Executive
In today’s pharmaceutical industry, it seems there are nearly as many communication channels as there are instruments in the Philharmonic.

Pharmaceutical Executive
Pharm Exec’s Brand of the Year selections from the last few years are still alive and kicking, although some have aged more gracefully than others.

Pharmaceutical Executive
Technology offers the promise of engaging patients on a level never seen before.

Pharmaceutical Executive
In the last three decades, MNCs have contributed greatly to the transfer of knowledge to generic companies in areas such as technology,

Pharmaceutical Executive
Every day, your competitors are doing everything within their power to steal your customers.

Pharmaceutical Executive
The three leading justifications for sticker-shocking drug prices in the US.

Pharmaceutical Executive
There has been a lot of concern about the decision to give the National Institute for Health and Care Excellence (NICE) the responsibility to look at “highly specialized technologies” (HSTs, or orphan drugs to you and me).

Pharmaceutical Executive
Global price management will lead the fight-back on margins, writes Arnaud Grunwald.

Pharmaceutical Executive
The European Medicines Agency thought it might at last be back on the road to salvation when Guido Rasi swept into town in 2011 and started ordering greater transparency in the agency’s operations.

Pharmaceutical Executive
Vaccine development is on a roll, boosted by biomedical research uncovering new molecular targets for preventives and treatments.

Pharmaceutical Executive
Human behavior may be the most significant variable in the future of cancer care in two sense.

Pharmaceutical Executive
At this year’s Bio IT World Conference, Thomas Verish, Group Director of Data Operations Services at Bristol-Myers Squibb,

Pharmaceutical Executive
Despite Mexico’s efforts to improve its regulatory framework and create a better business environment for pharma.

Pharmaceutical Executive
Several legal cases have challenged the Food and Drug Administration’s approach to regulating industry communications about medical products.

Pharmaceutical Executive
In anticipation of the 200th anniversary of the United States Pharmacopeia (USP) in 2020.

Pharmaceutical Executive
Novartis has teamed with the French drug maker Servier to develop and commercialize novel drug candidates targeting apoptosis in oncology.

Pharmaceutical Executive
A national publicity campaign recently succeeded in obtaining early access to an experimental treatment for a seriously ill child,

Pharmaceutical Executive
In light of the risks and challenges inherent in the development of oncologics.

Pharmaceutical Executive
It’s hardly news that biopharmaceutical companies today face increasing pressure to innovate, produce, improve efficiency and quality.

Pharmaceutical Executive
Pharm Exec’s Brand of the Year award is itself a brand with staying power - our first recipient was Merck’s Gardasil papillomavirus vaccine.

Pharmaceutical Executive
Europe’s pharma industry association EFPIA has launched its ”Manifesto for an Integrated Life Sciences Strategy in Europe”.