
Pharmaceutical Executive
As expected, the PFE/AZ saga has just begun to play out, and as we listen to the music of mega-mergers once again.

Pharmaceutical Executive
As expected, the PFE/AZ saga has just begun to play out, and as we listen to the music of mega-mergers once again.

Pharmaceutical Executive
Diabetes, it can be said, is one of the unpleasant side-effects of being human.

Pharmaceutical Executive
The Pfizer-AstraZeneca courtship raises many fascinating questions about the future shape of the international pharmaceutical industry.

Pharmaceutical Executive
A CCG had denied funding for oocyte preservation for a woman with Crohn’s disease before she had bone marrow transplant and chemotherapy.

Pharmaceutical Executive
In our final Q&A installment previewing next week’s iPharma2014 digital forum.

Pharmaceutical Executive
Gilead’s hepatitis C treatment Sovaldi will top the list of biggest selling drugs in 2020, according to UK market intelligence company, Evaluate.

Pharmaceutical Executive
More than half a century ago, way before anyone had dreamt up the notion of digital disruption.

Pharmaceutical Executive
The confirmation of Pfizer’s bid for an outright acquisition of AstraZeneca has drawn mixed reactions from the UK.

Pharmaceutical Executive
The dizzying flurry of press releases from both Pfizer Inc. and AstraZeneca over a possible PFE acquisition of AZN have Rx experts on both sides of the Atlantic buzzing.

Pharmaceutical Executive
The pancreatic cancer market is set to climb from $855.8 million in 2012 to approximately $1.21 billion by 2019 (a CAGR of 5.1%), says a report from GBI Research.

Pharmaceutical Executive
New data maintenance guidelines have sparked a flurry of activity in Europe.

Pharmaceutical Executive
The Incremental Cost Effectiveness Ratio (ICER) thresholds used by agencies such as the UK’s National Institute for Health and Care Excellence (NICE).

Pharmaceutical Executive
Gilead Silences has cleaned up on their new $1,000 Hepatitis C pill Sovaldi (84,000 per treatment), hauling in a record-breaking $2.3 billion in its first full quarter on the market.

Pharmaceutical Executive
The recently released Medicare data on payments to individual doctors doesn’t provide specifics on prescription drug outlays,

Pharmaceutical Executive
The Department of Veterans Affairs and other California panels’ solution to the new and extremely costly hepatitis (Hep) C drugs (Sovaldi and Olysio) is to reserve them for the patients with advanced liver diseases, including those awaiting transplants.
Pharmaceutical Executive
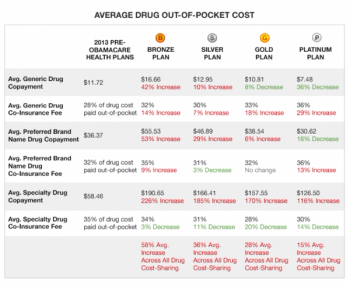
In the midst of the hue and cry last week over the public release of individual Medicare payments to physicians, I was surprised to read that one of Medicare’s most costly medical services is - ophthalmology.

Pharmaceutical Executive
Overall, last year can be considered quite a successful one in terms of new product approvals.

Pharmaceutical Executive
Health IT is going through an era of both expansion and convergence, in which the universe of available information is growing exponentially.

Pharmaceutical Executive
We spend a lot of time focusing on what our advertising says to physicians, what our reps say to physicians, and what our websites and various other channels communicate to patients.

Pharmaceutical Executive
The European Medicines Agency is determined to minimize opposition to its next moves on releasing clinical trial data.

Pharmaceutical Executive
Pharma has operated the same way for 100 years, but the US healthcare model has changed completely in the last 30 years, for payers, pharmacists, hospitals, and even at the clinician level.

Pharmaceutical Executive
It was recently said, in reference to the UK’s Value-Based Assessment(VBA) program, that innovation had not been “tried and tested in the pricing context”.

Pharmaceutical Executive
Few would argue with the desirability of innovation - something new, something that delivers value.

Pharmaceutical Executive
In response to the recent shooting at Fort Hood that killed four (including the killer’s suicide) and wounded 16, the mental health watchdog Citizens Commission on Human Rights (CCHR) views this and other recent incidents as a wake-up call to the dangers of antidepressants.

Pharmaceutical Executive
The new Internet Library from the European Patients’ Academy (EUPATI) will come online towards the end of 2015.
Pharmaceutical Executive
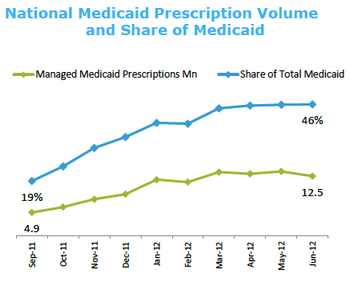
Cash-strapped states are shifting patients from traditional fee-for-service Medicaid coverage to managed Medicaid plans.

Pharmaceutical Executive
A report published yesterday provides recommendations for combating the “significant inequalities” affecting access to quality cancer care in Europe.

Pharmaceutical Executive
Monday’s decision by India’s Supreme Court to deny a patent for the top-selling oncologic drug Glivec took nearly a decade of litigation to resolve – but the implications in and beyond India are both immediate and lasting.

Pharmaceutical Executive
Since the early days of the internet, we’ve been busy creating websites and other digital channels.

Pharmaceutical Executive
The European Parliament yesterday voted towards the adoption of the new Clinical Trials Regulation.