Direct to Consumer: Moving House
These are demanding times for pharmaceutical marketers.
These are demanding times for pharmaceutical marketers. An already challenging regulatory environment has become downright hostile, as highlighted by some recent, dramatic actions:
» FDA's anti-DTC, conditional approval of Symlin (pramlintide) and Palladone (hydromorphone) has restricted the marketing of these life-changing drugs to consumers.
» In its heads up this spring, DDMAC indicated that it intends to advise not only on fair balance compliance, but on the subjective context of risk/side-effect presentation. (For example, DDMAC warned that a shot of a dog running in the park was "too distracting.")
» Comparative advertising has been effectively squashed as FDA came down on Crestor (rosuvastatin), for which AstraZeneca launched a campaign using comparisons to Pravachol (prevastatin), Zocor (simvastatin), and Lipitor (atorvastatin). Although DDMAC approved a chart that was used to support the claim, the superiority claim illustrated by the chart was ruled incorrect.
The result? Consumers are increasingly left with the impression that pharma marketers can't be trusted. Unfortunately, as an industry, we haven't done a particularly good job of gaining consumers' confidence. The problem is that we have been telling an incomplete story.

Ken Dec
Brands are the Currency of Trust
Rebuilding our fading trust with consumers will require a change in approach. People connect to brands to simplify decision-making about increasingly similar choices—choices they often make based on trust. Trust is the currency brands trade in, and to keep that value, we need to work on the relationship between consumers and pharmaceutical companies.
Continuing to invest almost exclusively in product advertising is no longer enough. Building meaningful trust requires communicating about and investing in the company brands that create these life-changing products. It also demands that we connect corporate brands more directly with product brands.
To take advantage of this opportunity, marketers must change their brand architecture model. (See "Brand Architecture Models,")
Play Offense, Not Defense
The "House of Brands" model—that is, a strong single-product brand identity without corporate branding—currently employed by industry is used primarily to manage two risks.
First, companies use it to manage drug performance risk. Marketers feel that if there are issues with a drug's market performance, safety, or efficacy, avoiding close corporate ties will minimize negative impact on the company's stock value.
Second, with a rapid rate of industry consolidation, firms are concerned that investments in corporate brands might disappear due to mergers.
Both of these defensive brand-management strategies are ineffective. As the COX-2 inhibitor cases and Biogen-Idec's withdrawal of the promising Tysabri (natalizumab) both demonstrate, Main Street and Wall Street know who is responsible for these drugs' performances and have punished them for their perceived shortcomings.

Merck and the controversy surrounding Vioxx (rofecoxib) shows the pitfalls of not investing in corporate brand building. We've heard the "collateral damage" argument that "it's a good thing Merck didn't connect its corporate brand to Vioxx more closely." The argument goes that Vioxx and other Merck product brands would be more seriously damaged if they were closely associated with the parent. That argument is seriously flawed from a brand-management and customer-experience perspective—in fact, the opposite is true.
The problem now is that, in the minds of many consumers and investors, Merck does equal Vioxx—and only Vioxx. If Merck had invested more in building their corporate brand—communicating its values and its mission—and connected it to its full line of products, including Fosamax (alendronate) and Vytorin (ezetimibe/simvastatin), the concerns would be limited to one of Merck's products, not with Merck as a company of committed, passionate people focused on creating healthier lives.
As for the industry consolidation defense of the House of Brands approach, this decision was always ill-conceived. As experienced brand managers in every category know, strong corporate brand investments raise the value of both the acquirer and the acquired; these brand investments are never wasted.
Put a Face to the Name
Companies should increase investment in the corporate brand significantly (even at the expense of shifting budget from product brand promotion) and move to the Branded House model, so that consumers truly understand pharma's mission: to improve the quality of people's lives. That means connecting product brands directly and obviously to the corporate brand. And not just as a logo afterthought, but the full Branded House model. That means marketing Pfizer Viagra (sildenafil) and Lilly Cialis (tadalafil).
There are numerous benefits to the Branded House approach:
» FDA can't touch corporate brand building.
» Each effort builds trust between consumers and pharma companies.
» The halo effect of corporate brand building will improve the effectiveness of our increasingly regulated product brand building.
» This approach generates a bigger bang for the buck for brand building efforts.
» It enables more differentiation by combining organizational and product differentiators in a powerfully combined message.
Without corporate brand building, a key communications element in building trust between our industry and consumers is missing. After all, in the same way that product brands can build trust in our products, pharma companies can build trust in our industry by increasing their focus on the message that we are a partner in patients' health, and not simply the peddlers of products. But without investing in corporate brands through our products and our industry, our bid to regain consumer trust will not work.
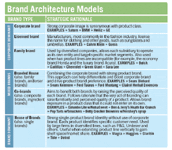
Brand Architecture Models
Merck's recent investment in promoting their patient-assistance program is a good start at building corporate brand value. But everyone in Big Pharma should be promoting these programs more effectively. And by doing so, each company's brand can then benefit from the goodwill created by educating consumers about these valuable programs.
The issues of the day demand a new DTC approach. Pharma companies can execute that change by moving from the House of Brands—which is a house of cards—to a Branded House.
Ken Dec is chief marketing officer at PreVision. He can be reached at kdec@previsionmarketing.com
People Jim Burke and Craig Douglass joined Medical Broadcast Company. Burke will be the new senior vice president, group management supervisor and Douglass will be the new senior vice president, group creative director.
» Kymberly Lee was named vice president of client development at Blue Diesel. » Ogilvy Healthworld named Angela Rossetti the new president of the company's New York advertising agency. » Stephen Sacchetti joined CommonHealth's Noesis as a senior vice president, account group supervisor. » DVC HealthCare announced Michele Cummins as the new vice president, account supervisor.

Kymberly Lee
Awards Novartis Pharmaceuticals Corporation and Deutsch, GlaxoSmithKline and Grey Worldwide, and GlaxoSmithKline and McCann Erickson won EFFIES in the health aids and prescription products category. UnitedHealthcare and GSD&M Advertising and BlueCross BlueShield of Western New York and Arnold Worldwide won EFFIES in the medical services category.

The Misinformation Maze: Navigating Public Health in the Digital Age
March 11th 2025Jennifer Butler, chief commercial officer of Pleio, discusses misinformation's threat to public health, where patients are turning for trustworthy health information, the industry's pivot to peer-to-patient strategies to educate patients, and more.
Navigating Distrust: Pharma in the Age of Social Media
February 18th 2025Ian Baer, Founder and CEO of Sooth, discusses how the growing distrust in social media will impact industry marketing strategies and the relationships between pharmaceutical companies and the patients they aim to serve. He also explains dark social, how to combat misinformation, closing the trust gap, and more.