
Joanna Huang analyses the use of external innovation tools among small and large pharma in China.

Joanna Huang analyses the use of external innovation tools among small and large pharma in China.

Pharmaceutical Executive
Despite a “temporary fix” at the ready, pharma supply repercussions loom if exit deal isn’t reached this month.

Pharmaceutical Executive
Dr. Lingshi Tan, a director at DIA, and chairman and CEO of Shanghai-based CRO dMed Company Limited, which he founded after establishing R&D operations for Pfizer in China, discusses the nation's regulatory evolution and opportunities for its government to strengthen collaborations with FDA and other leading global agencies.

Pharmaceutical Executive
How innovation growth has upped the pressure on China to compete globally, forcing upgrades in regulatory, quality, and standards.

Pharmaceutical Executive
How the nation’s rapidly changing regulatory climate is creating a new landscape for pharmaceutical service providers

Pharmaceutical Executive
Pharm Exec profiles Dr. Xiaobin Wu, an accomplished leader in helming China operations at big pharmas such as Pfizer and Bayer, who believes his recent jump to biotech as GM of China and president of BeiGene will deliver his most lasting legacy.

Pharmaceutical Executive
Green-lighting China as our coverage focus in the February issue wasn't without conflict or hesitation, but as our reporting of the nation's biopharma landscape shows, perseverance and growth many times go hand-in-hand.

Pharmaceutical Executive
Momentum and uncertainty shape the sector's economic picture.

What the new rules mean for Chinese-related investments and acquisitions in the US.

You can’t make progress without consensus. This holds true for Brexit and for the unparalleled opportunity that the WHO Roadmap report on Access to Medicines and Vaccines presents, writes IFPMA's Greg Perry.

“You wanna play rough? Just try it!” was the gist of the New-Year message from the European pharma industry to the healthcare authorities circling around drug firms preparing for pricing discussions. Reflector reports.

As we pass the 6-month anniversary of theGeneral Data Protection Regulation's effective date, the number of organizations which boast 100% GDPR compliance has certainly increased. But life sciences companies are still grappling with implementation of the policy and procedure changes, writes Ashley Williams.

James Clark addresses one of the key questions data protection and compliance officers are asking following the implementation of the General Data Protection Regulation -"What role am I playing under the GDPR?"

As the EU prepares for a change of governance, policymakers should take note of recent comments about the "poisoned atmosphere" surrounding healthcare policy debates, writes Reflector.

Whatever the final outcome of Brexit, investing in supporting advocacy communities will be an important element of maintaining trusted relationships with patient groups and stakeholders in the UK and the EU, write Nick Hicks and Tamsin Rose.

Pharmaceutical Executive
This article discusses three key considerations a manufacturer should either understand or seek a consulting partner to guide them through a commercialization plan.

The Commission’s gestures toward enacting formal standards to digitize health records are masking wider policy gaps.

Pharmaceutical Executive
Parliament throws wrench in Commission’s plan to regulate HTA.

Anjum Swaroop, Vice President and Head of IP at Dr. Reddy's, talks to Pharm Exec about her journey with the company and about what the future holds for this major US generics player.

Michael W. Magdycz share some of the lessons he's learned directing alliances spanning a range of countries and products.

Jin Zhang provides an overview of China's major bispecific antibody developers.

Disparities in income levels in China continue to drive far-reaching differences in how poorer and richer cancer patients are treated, with recent data suggesting that the differences in access to treatment lead to different outcomes for poorer patients. Peter De Richter reports.

Pharmaceutical Executive
What will come out of Austria’s shakeup of European pharma rules?

The call to reduce market entry times for life-saving new drug therapies is becoming more urgent. What constitutes best practice in managing translations in today’s regulatory affairs environment? Nancy Pollini reports.
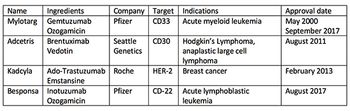
In the face of booming antibody market, China has jumped into the antibody-drug conjugate space. Jin Zhang reports.