
Pharmaceutical Executive
Over the last decade, Spain has seen a significant growth in venture-capital investment in the life sciences. We explore the driving forces.

Pharmaceutical Executive
Over the last decade, Spain has seen a significant growth in venture-capital investment in the life sciences. We explore the driving forces.

Research-based companies in Europe look as though they have lost one battle on preserving incentives for innovation – but the bigger war is only now getting underway, writes Reflector.



Artificial intelligence adoption by pharma companies in the UK demands a new, agile governance layer, write Tim Wright and Antony Bott.
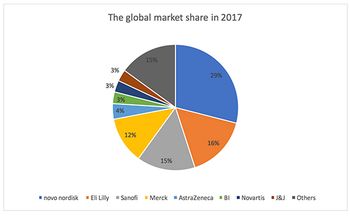
With China predicted to account for 48% of the global diabetes population by 2045, Jin Zhang looks at how the country's domestic pharma companies are faring in this treatment area.

Global pharma companies and Japan can create a win-win situation through a new pricing mechanism, writes Nobuko Kobayashi.

Raman Sehgal talks to three industry leaders for their insights on the challenges faced across the life science sector and how the EMA needs to respond.
Filip Conic outlines the market-access approaches and commercial strategies to unlock the growth potential in the Southeastern and Eastern European region.

Pharmaceutical Executive
Exploring the prospects-and related challenges-for Chinese life science as it aggressively pursues new growth areas.

Pharmaceutical Executive
New Jersey is still a top pick for companies when it comes to pharma.

Pharmaceutical Executive
Korean biotech opens first U.S. headquarters in New Jersey to better control clinical trials as it looks for strategic partnerships in the U.S.

Pharmaceutical Executive
Enzychem Lifesciences explains why the Korean biotech decided to open U.S. headquarters, as well as gives an update on their most recent clinical trials.

China's nurse educator program aims to improve quality of care and make life simpler for physicians handling high patient load. Sebastian Bather reports.

Figuring out the workings of the EU in topics related to health can be akin to deciphering Egyptian hieroglyphs, making it challenging for companies to plan ahead, writes Reflector.

On May 1, China eliminated the tariff on 28 categories of imported drugs. Jin Zhang looks at the implications for foreign and domestic pharma companies.
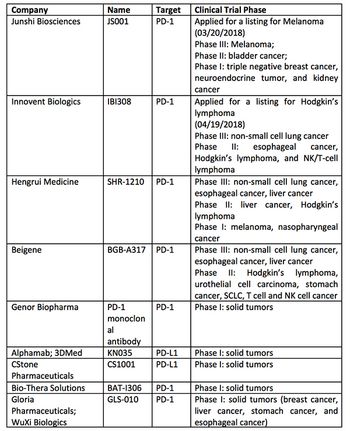
The PD-1/PD-L1 market in China is heating up and companies there have made a series of advances, writes Jin Zhang.

Pharmaceutical Executive
Pharm Exec’s 18th annual listing of the top biopharma players shows that most of the familiar placeholders are holding their ground as they weigh a changing business climate with new opportunities for growth in a potentially resurgent marketplace.

Pharmaceutical Executive
Pair of EU meetings, while well-meant, offered little action and beg the question-what’s the true merit in working together?

Innovations in Big Data will have a lasting impact on care and present an opportunity for U.S. pharmaceutical companies, writes Nobuko Kobayashi.
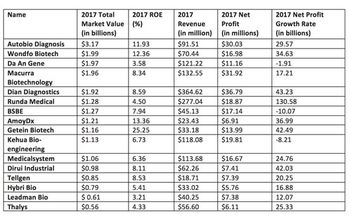
Jin Zhang identifies the major domestic companies in China’s IVD industry.

Jin Zhang discusses the research and development of several major biologics in China.

Jin Zhang looks at how China's CART-T therapy race is heating up.

Pharmaceutical Executive
Looking back at the agenda of a 1985 EFPIA conference demonstrates that, three decades on, the European drug industry is still seeking a pill for its own ills.

Pharmaceutical Executive
Outlining the four major areas driving China’s life sciences industry, which may be on the cusp of a new era in R&D and healthcare reform.