
Pharmaceutical Executive
Pharm Exec talks with David Dosa, a leading physician in geriatric medicine, whose famous work with a cat named oscar in end-of-life diagnostics became the stuff of therapeutic legend.

Pharmaceutical Executive
Pharm Exec talks with David Dosa, a leading physician in geriatric medicine, whose famous work with a cat named oscar in end-of-life diagnostics became the stuff of therapeutic legend.

As vital contributors to hospital disaster preparedness, the pharma industry has a role to play in making sure medical networks have what they need to meet the unforeseen, including future disasters.

Pharmaceutical Executive
Rare disease supporters deliver inspiration and optimism-as well as promise of regulatory cooperation.

Pharmaceutical Executive
The key steps to implementing the "patient access" model of the future

AmerisourceBergen believes the power of community begins with talk that gets the blood flowing.

A new IMS study reveals how changes in the design of commercial/employer-based health insurance plans are accelerating the push to higher patient contributions to the cost of prescribed drugs. William Looney reports.

As pharma continues to move towards "beyond the pill" strategies, better understanding of patients' sometimes paradoxical view of disease can help lead to strategies for helping them live more productive lives, writes Nick Hicks.

Nick Hicks looks at the challenges of devising a patient advocacy strategy amid the complex political, cultural and regulatory landscape of Russia and CIS.
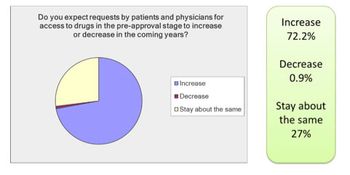
While the initiation and management of an access program can be challenging and requires careful assessment and planning, the benefits to patients, healthcare providers and sponsoring companies are immeasurable, writes Rob Fox. “Patients will find you.”In his keynote address at this year’s World Orphan Drug Congress, Henri Termeer, the former CEO, chairman and president of Genzyme reminded the audience that patients will actively seek out companies developing new therapeutics for their unmet medical needs.

Patient centricity requires clinical trials to be based on life outside the lab, writes Mads Holme.

Pharmaceutical Executive
Pharma’s image is still suffering despite a rise in medical innovation. Dan Bobear offers five patient-centered strategies that could help in finally mending the gap.

Emerging technology partnerships in the biopharma space may be provide hope in accelerating patient enrollment, writes Michael Christel.

Loreen M. Brown discusses how technology has influenced how healthcare teams function, from leveraging and delivering care to interacting with patients throughout the patient journey.

Casy McDonald looks forward to CBI's Patient Adherence and Access Summit in Philadelphia.

Pharmaceutical Executive
Gilead outlines its key access initiatives for its hepatitis C franchise.

Demands from patient advocacy groups for broader subgroup representation in clinical trials has generated a new drug trial transparency initiative at the Center for Drug Evaluation and Research (CDER).

Colleen Tracy James and Neil DuChez ask, is biosimilar litigation the new frontier as predicted or an area pursued less vigorously as under the Hatch-Waxman Act because of cost and manufacturing obstacles?

Steve Smith provides an advocates account of last month's Rare Disease Week on Capitol Hill.

Personalized medicine has reached a crossroads where fundamental change is not only appealing but essential, write Klarissa Hoday, Francesca Boggio, and Aleksander Ruzicic.

Research advocate, "cancer warrior" and survivor Jack Whelan talks to Pharm Exec about what pharma how pharma could improve its communication with patients.

Pharmaceutical Executive
Pharma jumps on board personal genomics train-at least for test drive-but will journey ultimately help transform treatment or stall out as just another fad?

Dr. Pamela Walker asks, in an increasingly competitive marketplace, with greater understanding of behavioral biases such as loss aversion, how can pharma professionals use loss aversion to propel brands to success?

Non-adherence, in the US alone, is a $100-billion-a-year problem. Healthcare players are touting patient education and engagement as the keys to better adherence rates. Ben Comer reports.

Elderly patients are underrepresented in clinical trials, but there are few obstacles to including this patient group in trials that cannot be overcome, writes Sydney Rubin.

Advocacy organizations and individual patients are getting more involved in every facet of the healthcare system, from drug R&D, to federal and state policy all fueled by the hour-to-hour passion of living with a disease.