
In the United States, the biomedical industry encounters numerous regulatory challenges in conducting nonclinical and clinical studies and managing chemistry, manufacturing, and controls (CMC) activities.

How the Continuing Need for Pain Medication Impacts the Pharma Industry

Treating Disruptive Symptom’s in Crohn’s Patients: Q&A with Mark Genovese, MD

In the United States, the biomedical industry encounters numerous regulatory challenges in conducting nonclinical and clinical studies and managing chemistry, manufacturing, and controls (CMC) activities.

Paul Howe, chief commercial officer at Protega Pharma, discusses how regulatory agencies are updating their policies based on the presence of abuse-deterrent medications.

The government agency says X no longer suits its communication needs.
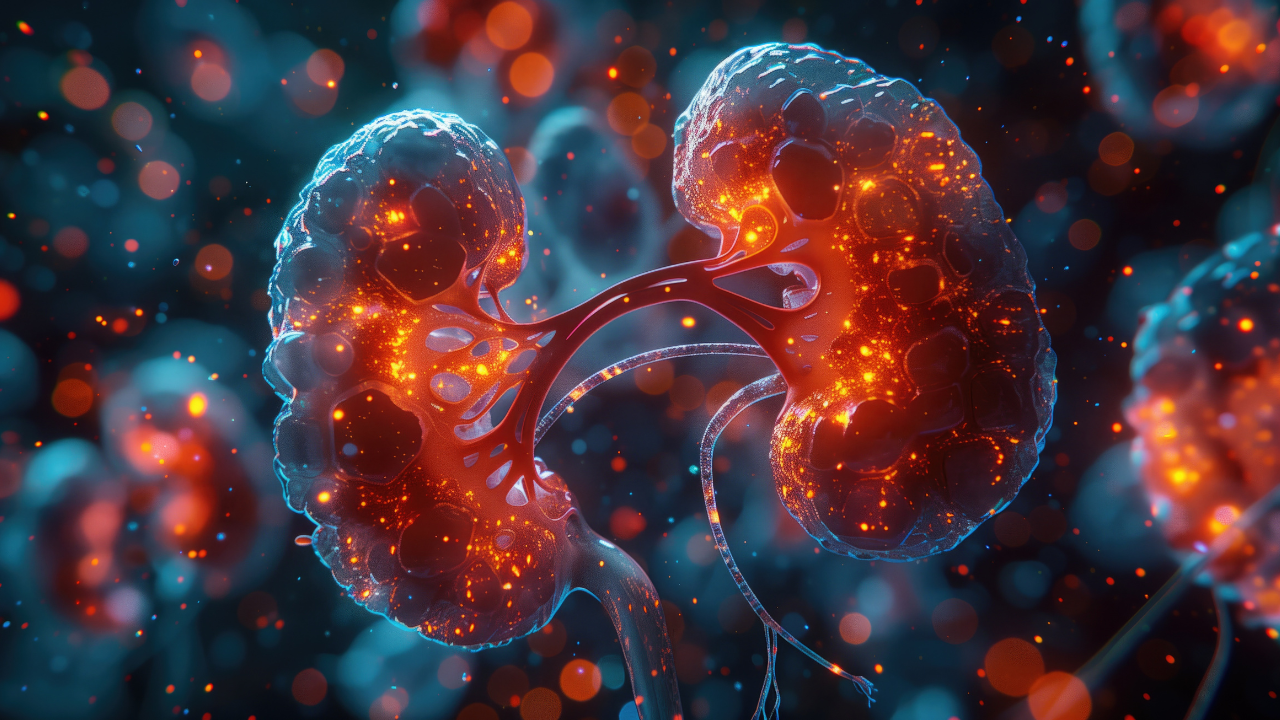
Priority review for Welireg is based on data from the Phase II LITESPARK-015 trial, which showed promising response rates in patients with advanced, unresectable, or metastatic pheochromocytoma and paraganglioma.
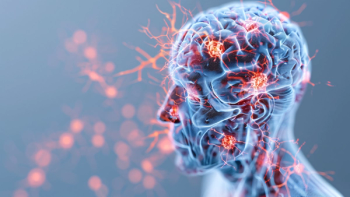
Approval was based on results from multiple studies, which demonstrated a slowing of cognitive decline in patients with Alzheimer disease administered Leqembi.

Application is supported by results from the DEVOTE study, which suggested that two doses of Spinraza (nusinerse) 50 mg taken 14 days apart can deliver significant clinical benefits in patients with spinal muscular atrophy.

As part of the regulatory action, memantine/donepezil extended-release capsules have been granted a 180-day exclusivity in patients with Alzheimer disease, with Everolimus receiving approval for tuberous sclerosis complex.
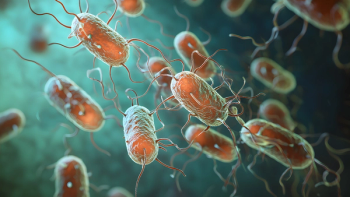
The cobas liat test aims to improve patient outcomes by offering rapid, accurate results in decentralized and community-based settings for patients at risk for chlamydia, gonorrhea, and Mycoplasma genitalium.

Spravato is the first and only monotherapy to gain FDA approval for treatment-resistant depression.

Approval is based on results from the TROPION-Breast01 Phase III trial, in which Datroway demonstrated a significant reduction in the risk of disease progression or death in patients with previously treated, unresectable, or metastatic HR-positive, HER2-negative breast cancer.
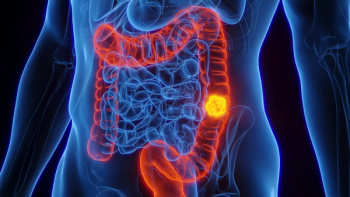
Approval is based on data from the Phase III CodeBreaK 300 trial, which demonstrated that treatment with Lumakras and Vectibix significantly improved progression-free survival in patients with KRAS G12C-mutated metastatic colorectal cancer.
Approval follows results from the Phase III ECHO trial, which demonstrated that Calquence plus chemotherapy reduced the risk of disease progression or death by 27% in patients with previously untreated mantle cell lymphoma.
Niktimvo marks the first FDA-approved therapy targeting CSF-1R to address inflammation and fibrosis in chronic graft-versus-host disease.
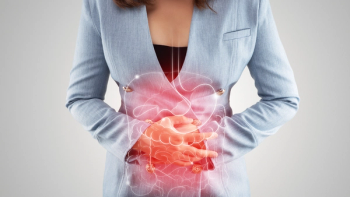
Approval was based on results of the VIVID-1 study, which found that Omvoh demonstrated significant clinical remission and endoscopic response rates in patients with Crohn disease.
The New Drug Application for TAR-200 is supported by results from the Phase IIb SunRISe-1 study, which demonstrated an 83.5% complete response in patients with Bacillus Calmette-Guérin-unresponsive high-risk non-muscle-invasive bladder cancer.

Acceptance of the Biologics License Application for subcutaneous Leqembi is based on results from the Clarity AD open-label extension and modeling of observed data in patients with Alzheimer disease.
Priority Review was based on results from the Phase III Vivacity-MG3 study of nipocalimab in patients with generalized myasthenia gravis.
Abrysvo and Arexvy will now be required to come with labeling that includes a warning about a potential increased risk of Guillain-Barré Syndrome.
Fast Track designation was based on results from a Phase II study, which demonstrated that VGT-309 was safe, well-tolerated, and enhanced tumor visualization in lung cancer patients.
Breakthrough Therapy Designation for GSK5764227 was based on data from the ARTEMIS-002 Phase II trial, which demonstrated promising efficacy and safety in 42 osteosarcoma patients.
The designation marks VGA039 as the first drug candidate to receive FDA Fast Track designation for von Willebrand disease.
Approval of Tevimbra was supported by the RATIONALE-305 Phase III trial, which demonstrated a significant overall survival benefit in patients with unresectable or metastatic HER2-negative gastric or gastroesophageal junction adenocarcinoma.
Approval was based on results from the Phase III CheckMate-67T trial, which demonstrated non-inferior pharmacokinetics compared to intravenous Opvido.

In this part of his Pharmaceutical Executive video interview, Peter Ax, CEO of UpScriptHealth, discusses regulatory challenges and opportunities that telehealth companies face in 2025.

Approval marks the first generic version of liraglutide injection, referencing Victoza, for improving glycemic control in adults and children aged 10 years and older with type 2 diabetes, alongside diet and exercise.