Pharmaceutical Executive
Value-based pricing in the United Kingdom is getting closer, but it remains out of focus.
Pharmaceutical Executive
Value-based pricing in the United Kingdom is getting closer, but it remains out of focus.

Legal battles and regulatory missteps undermine access to low-cost generics, at home and abroad, writes Jill Wechsler.

Pharmaceutical Executive
The European Union's attempt to update its transparency rules sparks new debate.

Pharmaceutical Executive
There seems no end to demands for data on clinical research, conflicts of interest, company payments, and drug prices.

Pharmaceutical Executive
European Commision blocks the authorization of a life-saving liver drug outside of France.

After a decade of strife, the dialogue between industry and government appears to have entered a positive new phase. Julian Upton reports.

Compliance officers have risen into management's highest ranks, by choice in some organizations, and by government decree in others. Either way, their importance as a strategic partner can hardly be understated.

Pharmaceutical Executive
FDA policies will reshape drug development, while pressure to cut healthcare spending may alter drug coverage and pricing.

Pharmaceutical Executive
If people can promote drugs for uses that lack supporting evidence, it would turn back the clock to the pre-1962 world of medicine where there wasn’t any research or data on what medicines worked and what was harmful, says Robert Temple, deputy director for clinical science at FDA’s Center for Drug Evaluation and Research (CDER).

Pharmaceutical Executive
Increased scrutiny and new regulation have seen pharma’s relationship with key opinion leaders (KOLs) undergo a paradigm shift in recent years.

Pharmaceutical Executive
Tax and budget decisions will shape the healthcare market and drug research and regulation.
Pharmaceutical Executive
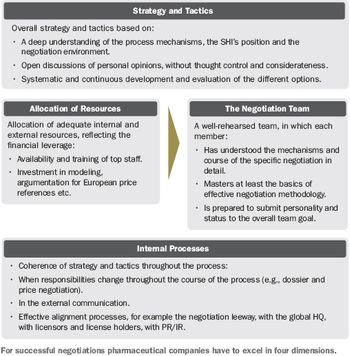
At two years and counting, what's the secret of success in negotiating pricing under the new AMNOG legislation?

Pharmaceutical Executive
Europe's chief regulator charts a strategy from many moving parts. Dr Guido Rasi talks to Pharm Exec's William Looney.

Pharmaceutical Executive
Recent change likely to boost drug coverage by plans, but with variation in formularies.

Pharmaceutical Executive
Over-the-counter products face a new wave of regulation.
Pharmaceutical Executive
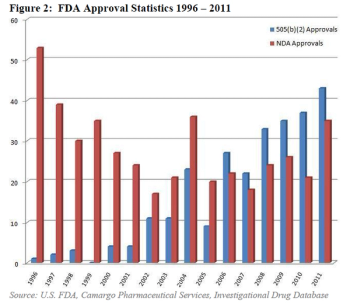
It was in 1984 when FDA first added the 505(b)(2) pathway for drug approval, a hybrid between the accelerated pathway for generic drug applications, and the standard de novo NDA pathway for proprietary drugs.

Pharmaceutical Executive
With the Supreme Court’s decision in June to uphold the Patient Protection and Affordable Care Act (ACA), life science companies gained access to 32 million potential new customers.

The new head of EFPIA faces a Promethean challenge: selling the merits of costly science and innovation in an era of bristling competition, fiscal crisis, and declining demographics.

Pharmaceutical Executive
The most obvious competitors for Sanofi’s Aubagio, the second oral multiple sclerosis (MS) drug to receive FDA approval, would appear to be Novartis’ Gilenya.

Pharmaceutical Executive
Europe's fiscal crisis is becoming a new rallying cry for patient groups-but regional unity is still elusive.

Pharmaceutical Executive
Bending the cost curve back to valuing the cycle of life.

Pharmaceutical Executive
Well, we certainly have a presidential campaign now, don’t we? Good grief. Since Rep. Paul Ryan was named the Republican VP candidate last week.

Comparative research documenting value and affordability is key to obtaining coverage for high-cost therapies. Jill Wechsler reports.

Pharmaceutical Executive
It’s back to business for pharma and biotech companies. The main result of the Supreme Court decision upholding the Affordable Care Act (ACA) is that it ends all the rampant speculation and uncertainty about the future shape of the U.S. healthcare system.

So, it looks like June 25th will be the moment of truth we have all been anticipating. The Supreme Court is expected to hand down its ruling on the 2010 healthcare reform law. Who knows what will happen?