
Pharmaceutical Executive
The FDA proposes to use the RWE Program to guide generation of data in support of approval for new indications or to help support post-approval study requirements.

Pharmaceutical Executive
The FDA proposes to use the RWE Program to guide generation of data in support of approval for new indications or to help support post-approval study requirements.

FDA restarted full operations on January 28 following Congressional agreement on a continuing resolution to fund government operations for three weeks. However, the situation will get much worse if Congress and the White House fail to negotiate a longer-term budget for 2019 by February 15.

It’s been a banner year in the new drug approval business: CDER approved 59 novel drugs and biologics in 2018, surpassing previous high marks and improving on recent gains, with additional new products approved by the CBER boosting the total.

Pharmaceutical Executive
CDER priorities for 2019 include opioids, quality, safety, and innovation.

As we pass the 6-month anniversary of theGeneral Data Protection Regulation's effective date, the number of organizations which boast 100% GDPR compliance has certainly increased. But life sciences companies are still grappling with implementation of the policy and procedure changes, writes Ashley Williams.

James Clark addresses one of the key questions data protection and compliance officers are asking following the implementation of the General Data Protection Regulation -"What role am I playing under the GDPR?"

Pharmaceutical regulatory divisions should use the same discipline, rigor and focus on the talent pipeline as they do the product pipeline, writes Karin Van Baelen.

The industry should take a twin-track approach to the potential threat of the Trump administration's proposed move to reference pricing, writes Todd Edgar.

Despite ongoing efforts to address the problem, FDA now sees a rise in active shortages of important medicines and in the duration of supply problems. Jill Wechsler reports.

Pharmaceutical Executive
Regulators, prosecutors shifting enforcement focus from off-label promotion to messages that raise safety issues, mislead.

Healthcare was the deciding issue for more than 40 percent of voters in the mid-term elections. Despite disagreement on most of the issues, leaders of the two parties are open to challenging the status quo on drug costs and spending.

President Trump's International Pricing Index proposals amount to international reference pricing (IRP), something that countries in Europe have been doing for years. Leela Barham asks, What lessons can be learned from there?

FDA's proposal to build a modern system to gather real-world evidence (RWE) from about 10 million individuals could have profound implications, but it will realize its potential only if FDA takes a more expansive view of what RWE can be, writes Paul Glimcher.

The Regenerative Medicine Advanced Therapies (RMAT) designation was introduced by the 21st Century Cures Act as a pathway to accelerate FDA approval and market entry of regenerative medical therapies. Two years on, William Rose and Suchira Ghosh identify the trends in how FDA assesses the clinical evidence supporting successful RMAT candidates.

Pharma says new "free-market" plan proposed by President Trump and HHS will harm patients. Jill Wechsler reports.
Pharma companies have faced hurdles in gaining approval of Janus kinase (JAK) inhibitors for the treatment of rheumatoid arthritis. Denise Baldock, Elizabeth Baynton, Amanda Baskett, and Nicola Bailey look at what can be learned from the lessons of the past.

In publishing its proposed rule on disclosing drug prices just three weeks before the mid-term Congressional elections, it’s not a big stretch to see the move by the Trump administration as designed for short-term political purposes.
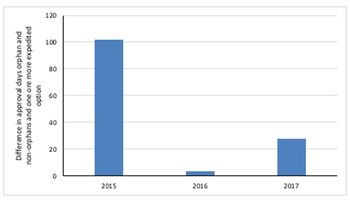
Leela Barham looks at whether there is a boost to the speed of FDA approval when a drug not only secures one or more of the FDA’s expedited development and review methods, but also when they are also designated an orphan drug.
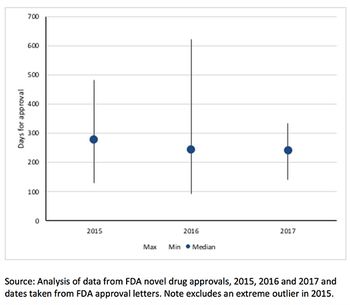
There’s been a lot of talk about speeding up drug approval at FDA. So just how fast can approval be? Leela Barham takes a look at the speed of approval for each of FDA's expedited development and review methods.
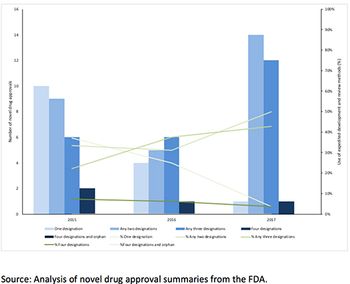
Leela Barham takes a look at the take-up of the existing options to speed up FDA approval.
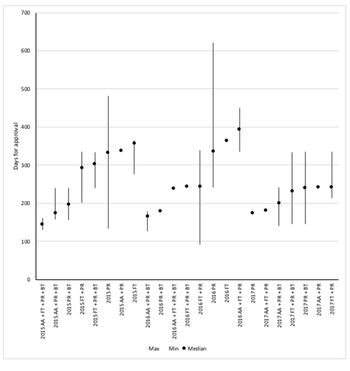
FDA offers four options to speed up approval and they can be used in combination. But just which of the many combinations offers the fastest approval? Leela Barham reports.


The call to reduce market entry times for life-saving new drug therapies is becoming more urgent. What constitutes best practice in managing translations in today’s regulatory affairs environment? Nancy Pollini reports.

CDER director plans to start implementation of new review practices by the end of 2018.

FDA is joining with other federal health agencies and the biomedical research community to advance the science, regulatory policies and reimbursement strategies to support innovative new medicines to combat infectious diseases.