
Alan White explores alternative ways for life sciences firms to keep hold of experienced, qualified professionals as demand for pharmacovigilance skills soars.

Alan White explores alternative ways for life sciences firms to keep hold of experienced, qualified professionals as demand for pharmacovigilance skills soars.

Having invested heavily in new regulatory database systems, life sciences firms owe it to themselves to capitalize on the insights locked within those rich data assets, writes David Gwyn.

Top of CDER’s to-do list for 2020 is tracking adverse events more effectively and combating the opioid crisis.

Goals for IDMP in Europe must not be diluted in 2020, if standardized medicinal product data is to be of tangible real-world benefit, says Remco Munnik.

Pharmaceutical Executive
Challenges will mount to established models for researching, developing, and marketing new therapies.

What to make of new momentum in advancing health agenda.

While not setting any records for the rapid approval of new drugs in 2019, FDA did speed a number of important new therapies to patients, writes Jill Wechsler.

Pharmaceutical Executive
Sponsors developed important new therapies amidst ongoing concerns over drug quality and costs.

While labor and tariff reforms in the revised North American trade agreement may have more visible impacts on the United States economy, the final document levels a major blow to exclusivity and patent protections important to innovator biotech and pharma companies.

Jill Wechsler looks at the CDER Office of New Drugs' new structure, which features more operational support for review functions and closer alignment of review offices to therapeutic categories.
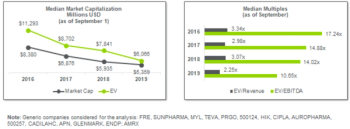
Pharmaceutical Executive
As the market for specialty and generic products continues to become more competitive and pricing pressures increase, the 505(b)(2) pathway may allow companies more options to diversify their portfolios.

Graham Francis sets out 5 critical business drivers for building a global enterprise labelling strategy over the year ahead.

A NASEM report stresses the importance of information sharing by biopharma companies and cooperation among regulatory authorities.

2019 saw progress towards ISO Identification of Medicinal Products data standards implementation, but making patients front and central to project focus must remain a priority, says Frits Stulp.

Developers of biosimilars have become dismayed with difficulties in gaining acceptance and reimbursement from the US healthcare system.

CDER's Rare Disease Cures Accelerator initiative looks to foster a coordinated research approach and methods that can expedite development of drugs to treat some of the 7,000 rare diseases.

A new drug shortages report from FDA revives the call for a rating system to help drug purchasers, hospital buying groups and consumers identify quality products that warrant higher prices. Jill Wechsler reports.

Pharmaceutical Executive
Coexisting with the DSCSA may hinder new legislative efforts.

Pharmaceutical Executive
Lapse spotlights wider actions to ensure data accuracy.

The Center for Drug Evaluation and Research has finally gained approval for broad changes in its process and procedures for evaluating and approving new drugs.

Time is running out on Ned Sharpless’ term as FDA acting commissioner, generating much talk as the administration shows interest in naming a new head for the agency.

Pharmaceutical Executive
Market consolidation limits sales, while legal challenges offset regulatory gains.

As part of the legislation expanding the federal Open Payments program, drug and medical device manufacturers will have to report payments and other “transfers of value” to a broader range of health professionals.

Jill Wechsler casts her eye over the multitude of health reform measures emerging on Capitol Hill.

It is worth keeping patients, clinicians, pharmacists and the wider public front of mind when evaluating priorities and best next steps towards Identification of Medicinal Products(IMDP) and other emerging standards, writes Frits Stulp.