
Pharmaceutical Executive
Agency focused on advancing testing and production methods, seeking input from other regions on common approaches.

Every big shift will stir up dissenters, but these negative forces can actually offer a useful contribution if their concerns are fed into a continuous change program. This could make all the difference in regulatory information management transformation, writes Steve Gens.

Pharmaceutical Executive
Agency focused on advancing testing and production methods, seeking input from other regions on common approaches.

In their latest article summarizing efforts to put each of the “American Patients First” blueprint's four strategies into action, Rick Kelly and Nisha Desai focus on the Lowering Out of Pocket (OOP) Costs strategy.

Pharmaceutical Executive
How R&D organizations can leverage FDA’s final guidance on pre-approval information exchange (PIE) to engage with payers prior to approval and launch.

What to make of the simmering public and political angst

Pharmaceutical Executive
New guidances open the door to interchangeables and less costly insulin.

In the third in a series of articles summarizing efforts to put each of the “American Patients First” blueprint's four strategies into action, Rick Kelly and Nisha Desai focus on “Incentivizing Lower List Prices.”

R&D organizations can leverage FDA’s final guidance on pre-approval information exchange, otherwise known as PIE, to engage with payers prior to regulatory approval and commercial launch.

A provision in a Senate health reform bill has reignited debate over the whether biological products should have to meet product quality standards established by the U.S. Pharmacopeia.

Congress is weighing in on drug pricing with a range of measures that differ in style and substance.

Pharmaceutical Executive
Cell and gene therapies in crosshairs of pricing focus, prompting stepped-up proposals on ways to finance these products.
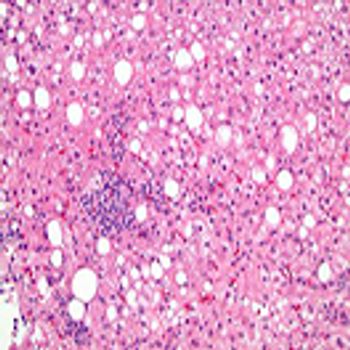
Sabina Heinz and Elizabeth Baynton look at how the continued absence of approved products affecting the management of NASH patients.

In the first in a series of articles addressing each of the American Patients First blueprint 's four strategies, Rick Kelly and Nisha Desai focus on “Increasing Competition”.

Jill Wechsler looks at the proposed new FDA rules to beef up regulatory resources and promote digital technologies to support the development and marketing of safe and innovative over-the-counter therapies.

FDA is examining and updating its programs for overseeing global operations and international affairs.

Pharmaceutical Executive
New leadership faces a host of complex initiatives to promote innovation and protect public health.

Elvis Paćelat outlines some lessons from big pharma as medical device manufacturers succumb to new reporting obligations.

New FDA leader Ned Sharpless will seek further solutions to the opioid crisis and work to reduce cigarette use in adults and kids.

A new CDER “knowledge management” approach will see companies submit applications that can be transmitted to experts from multiple disciplines able to assess applications for new drugs and biologics in a timely and efficient manner.

Pharmaceutical Executive
Follow-on product naming, messaging challenge FDA, industry.

Europe slow to action on emerging technologies, but the complex issues clouding EHRs and gene editing, for example, offer pause.

Pharmaceutical Executive
Executives from DIA, Ranjini Prithviraj and Sudip Parikh discuss documents the FDA released that further detail the agency’s implementation plan for its Software Precertification Pilot Program.

Pharmaceutical Executive
The digitization of healthcare is paving the way to accelerate drug development in the industry, and the FDA is doing its best to support industry.

Pharmaceutical Executive
Wide-ranging probes to target marketing, pricing, and access.

Gottlieb’s tenure included record new drug approvals and steps taken to curb opioid abuse.

Pharmaceutical Executive
Dr. Lingshi Tan, a director at DIA, and chairman and CEO of Shanghai-based CRO dMed Company Limited, which he founded after establishing R&D operations for Pfizer in China, discusses the nation's regulatory evolution and opportunities for its government to strengthen collaborations with FDA and other leading global agencies.