
Pharmaceutical Executive
Instead of developing innovative therapies, pharma companies devote most of their resources to modifying existing medications, according to a report from the National Institute for Health Care Management Foundation (NIHCM).

Pharmaceutical Executive
Instead of developing innovative therapies, pharma companies devote most of their resources to modifying existing medications, according to a report from the National Institute for Health Care Management Foundation (NIHCM).

Pharmaceutical Executive
Many more high-risk patients could benefit from statin therapy than currently receive the cholesterol-lowering drugs, according to a major UK study set up by the Medical Research Council and the British Heart Foundation.

Pharmaceutical Executive
Pharmaceutical companies and their marketing partners face a common dilemma: how to translate scientific progress into market success. Despite huge investments, R&D never seems to produce an adequate number of products with blockbuster potential.

Pharmaceutical Executive
A CenterWatch survey of 545 European clinical trial investigators named the best pharma companies to work with in clinical development.

Pharmaceutical Executive
Shares in Danish company Novo Nordisk took a hit after it suspended trials for one of its main pipeline candidates, the dual-acting insulin sensitizer, ragaglitazar.

Pharmaceutical Executive
Spurred by concerns about rising drug expenditures, the Netherlands' government has introduced a list of "lifestyle" medicines that it intends to stop covering with its basic health insurance.

Pharmaceutical Executive
In a new twist on "direct mail campaigns," marketers at Eli Lilly have taken DTC to an unprecedented level. The company is currently under investigation by the Florida attorney general for allegedly mailing Prozac Weekly (fluoxetine) to a woman who did not have, or request, a prescription for the product.
Pharmaceutical Executive
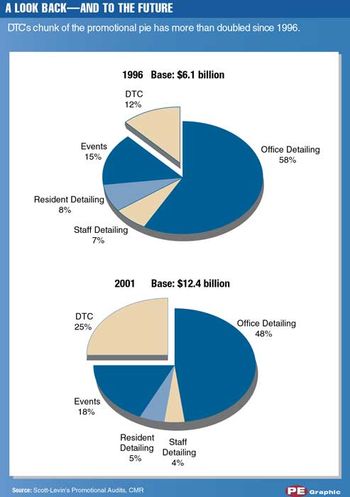
Pharma companies invested a hefty $12 billion in promoting to physicians and consumers in 2001, according to Scott-Levin. But after the highs of 2000, spending in 2001 seemed stunted-a growth rate of 9 versus 13 percent.

Pharmaceutical Executive
Sixteen years ago I began to edit Pharmaceutical Executive, a magazine unknown to me only a few months before. Today, I wonder how much I've learned in all the time since.

Pharmaceutical Executive
The number of pharmaceuticals derived from nature is staggering. Many medicine cabinet staples were first isolated from natural sources-penicillin from mold, quinine from the cinchona tree, digitalis (foxglove) for heart ailments, and the list goes on.

Pharmaceutical Executive
The Enron/Arthur Andersen debacle has been a painful warning that there is more to analyzing corporate performance than the smoke and mirrors that often pass for accounting and financial scrutiny. Indicators such as brand value, new product revenue, presence and percentage of business conducted in the US market, and sales growth provide more reliable criteria for companies' short and long-term prospects.