
Amid the limitations of traditional compliance programs that emphasize control measures, here are steps biopharma companies can implement in their own cultures to ensure they’re behaving and performing with integrity.

Amid the limitations of traditional compliance programs that emphasize control measures, here are steps biopharma companies can implement in their own cultures to ensure they’re behaving and performing with integrity.
A recent roundtable of pharmaceutical CEOs, and moderated by Dr. Scott Gottlieb, discussed pressing concerns around the pandemic, including clinical trials, product launches, keeping employees safe and the future of the pharmaceutical industry.

While COVID-19 has shifted the rules of the healthcare industry, research shows there was already progress on changes being made for how pharma companies are launching products.

With the role of Medical Information in pharma companies changing in recent years, this article outlines some current and future trends including globalization and the use of data to drive change.
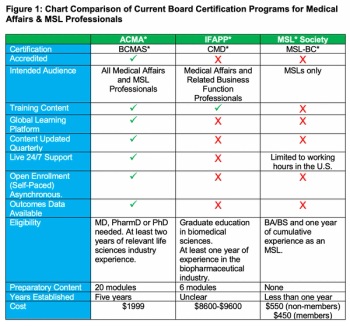
With the growth of the medical science liaison (MSL) role in mind, this article provides a comprehensive review of the board certification programs that are currently available to MSLs and other medical affairs professionals.
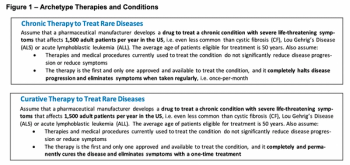
There has been a considerable amount of attention given to the rising costs of pharmaceutical and biotechnology therapies in the US. In early 2020, Charles River Associates conducted this research to determine what "fair" prices would be for these therapies.
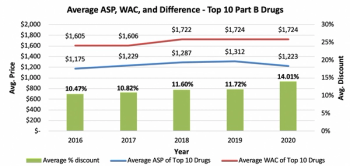
While there has been an increase in discourse concerning drug pricing transparency in recent years, there is still a degree of misunderstanding. This is due to the healthcare industry's numerous stakeholders and their compelling interests. This research done by Guidehouse aims to clear the picture.

As the number of biologics facing patent expiration in the US continues to rise, the market is further opening up to biosimilar manufacturers. George I'ons offers some pointers on how to secure an attractive market share during this time.

How exactly do pharma companies account for a gap between their hopes for a new drug and reality? Peter Harbin reports.
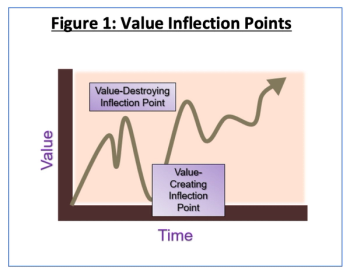
Dealing with significant unplanned events in alliances is important work. It's is critical to maintain the ongoing health of the alliance by applying sound governance principles and minimizing the disruption to normal operations wherever possible. So how can you stabilize your ships in rough seas?

Across all pharma companies' activities, the COVID-19 crisis has reinforced the need for pharma leaders to commit to, communicate, and exemplify the purpose that is at the center of their strategies and organizations.

Bill Barr outlines the systematic application of three criteria to guide discounting decisions.

Donna L. LaVoie discusses four key industry areas that will experience downstream pressures in the wake of the COVID-19 crisis.
In 2020 and beyond, succeeding in the new ecosystem means focusing on delivering value backed by data, tailored engagements with a diverse range of distinctly defined stakeholders, and monitoring the rapidly evolving healthcare space for new trends that will impact success.

Fully understanding the construct of what it means to be lean can equip individual contributors, teams, and leaders to contribute to the fullest extent of their capabilities.

Pharmaceutical Executive
Understanding how CIOs can overcome the challenges of building a digital health Internet of Things (IoT) infrastructure is essential for harnessing digital transformation in a cost-effective and scalable manner.

Pharmaceutical Executive
Eight lessons to drive better outsourcing decisions.

Pharmaceutical Executive
Five key areas biopharma manufacturers focus on to predict where the markets will be and determine how standards of care will evolve to effectively position new products.

Mike Kean and Billy Tamulynas discuss the key components of growth-ready technology for early-stage pharma companies and how to make optimal use of them.
To ensure patients have timely access to expensive new therapies, biopharma companies have to address commercialization challenges with innovative measures, even after getting regulatory approval.

A key question for an organization post-M&A is: “What organizational capabilities support global guidance and leadership, and which of them require these at the 'local' level?” Scott Hull discusses potential solutions.

While culture may "eat" strategy, shared understanding creates both culture and strategy, write Steve Figman and Sean Robertson.
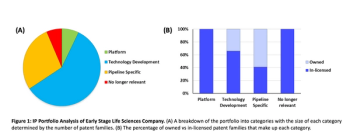
Pharmaceutical Executive
Creating an IP portfolio strategy that includes R&D, in-licensing, out-licensing, and mergers and acquisitions can help businesses stay ahead of the competition.
Pharmaceutical Executive
Specialty pharmacy's role in shifting the existing paradigm from a transactional model to an experiential model in ways that can fuel real-world data acquisition and strategy.

Jianan Huang discusses how the drug revenue formula is being used to guide R&D "rescue strategies".