
The latest news for pharma industry insiders.

The latest news for pharma industry insiders.

Artiva Biotherapeutics' CEO Fred Aslan, MD, discusses the concept of an immune reset and what the company is tracking in trials to assess its potential.

March is Colorectal Cancer Awareness Month, and the health plan hopes to improve survival rates with early screenings.
Sun Pharma’s acquisition of Checkpoint Therapeutics includes Unloxcyt, an FDA-approved anti-PD-L1 therapy for metastatic and locally advanced cutaneous squamous cell carcinoma.
Omlyclo, a biosimilar to Xolair, is indicated for the treatment of moderate to severe persistent asthma, chronic rhinosinusitis with nasal polyps, immunoglobulin E-mediated food allergies, and chronic spontaneous urticaria.

Jonathon Whitton, AuD, PhD, VP, auditory global program head, Regeneron, discusses how key regulatory designations, such as the FDA's Regenerative Medicine Advanced Therapy designation, facilitate accelerated development and commercialization of DB-OTO through increased collaboration with regulators.

Artiva Biotherapeutics' CEO Fred Aslan, MD, discusses two ongoing trials for autoimmunity indications in the US and how AlloNK differs from traditional B-cell depletion strategies.

The latest news for pharma industry insiders.
Five-year data from Phase III trials demonstrated that 67.7% of patients moderate-to-severe plaque psoriasis treated with UCB’s Bimzelx achieved complete skin clearance.

Clinical trial results found that a majority of patients with atopic dermatitis who were treated with Ebgylss achieved complete or near complete skin clearance at three years on a single monthly maintenance dose.

Jonathon Whitton, AuD, PhD, VP, auditory global program head, Regeneron, discusses the promising results of DB-OTO in the CHORD trial for children with otoferlin-related hearing loss.

The data analytics company based its new product on simple spreadsheet technology.

The latest news for pharma industry insiders.
The approval of neffy marks the first major advancement in epinephrine delivery for patients over four years of age in more than 35 years.
Joint venture is expected to leverage Astellas' expertise in cell therapy R&D and Yaskawa’s robotics technology to enhance accuracy, reproducibility, and efficiency in cell manufacturing.

The latest news for pharma industry insiders.

In this Pharmaceutical Executive Video Interview, Peter Ax, Founder & CEO of UpScriptHealth, describes how his company's recent partnership with Pfizer is impacting drug access.

The acquisition is expected to enhance Jazz's oncology portfolio by adding dordaviprone, a novel small molecule therapy in development for H3 K27M-mutant diffuse glioma.
Submission is supported by positive results from the Phase III REGENCY trial, which demonstrated that nearly half of patients with lupus nephritis who received Gazyva/Gazyvaro plus standard therapy achieved a complete renal response.

The latest news for pharma industry insiders.
New indication for Tevimbra in combination with platinum-containing chemotherapy as a first-line treatment addresses an unmet need for adults with unresectable or metastatic esophageal squamous cell carcinoma whose tumors express PD-L1.
Stoboclo, a biosimilar to Prolia, is indicated for postmenopausal women and men at high risk of fracture, while Osenvelt, a biosimilar to Xgeva, is indicated for preventing skeletal-related events in patients with multiple myeloma and bone metastases from solid tumors.

Aliya Omer, VP, US franchise head, breast cancer, AstraZeneca, discusses how results of the trial compare with other treatments for gBRCAm HER2-negative high-risk early breast cancer.

The latest news for pharma industry insiders.
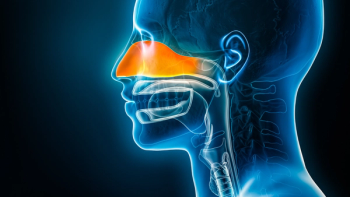
In the SWIFT and ANCHOR trials, depemokimab demonstrated statistically significant reductions in nasal polyp size, obstruction, and asthma exacerbations compared to placebo.

Under terms of the deal, Gubra will receive an initial $350 million payment and could earn up to $1.875 billion more in development and sales milestones for the potential weight loss drug.

Ron Lanton, Partner, Lanton Law, discusses the legal and industry challenges posed by recent National Institute of Health budget cuts, diversity bans in clinical trials, and pharmaceutical supply chain disruptions due to tariffs.

The latest news for pharma industry insiders.

Three of the new sites will focus primarily on producing ingredients for domestic usage.
FP008 is designed for patients with solid tumors that have not responded to existing immunotherapies targeting PD-1.