
Pharmaceutical Executive
The UK coalition government's consultation on the intro of value-based pricing has invited caution and resistance

Pharmaceutical Executive
The UK coalition government's consultation on the intro of value-based pricing has invited caution and resistance

Pharmaceutical Executive
The EU is largely powerless when it comes to policies over pricing and reimbursement
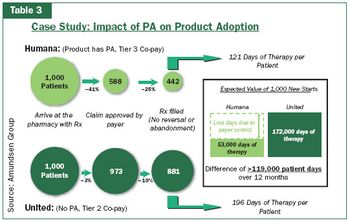
Pharmaceutical Executive
The pharmaceutical industry needs to use new and better data to accurately measure how much it is willing to invest in avoiding plan control

Pharmaceutical Executive
In France, it took a president's keynote speech to underscore the government's new understanding that there is more to pharmaceuticals than cost-containment

Philippa Montgomerie outlines the implications of the Court of Justice of the European Union's recent patent litigation forum shopping.
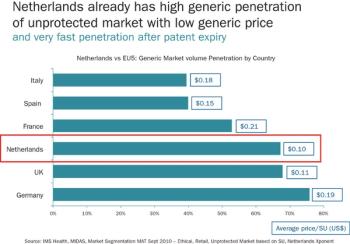
Pharmaceutical Executive
Despite its dimunitive size, the pharmaceutical industry looks to the Netherlands for a world-class medical infrastructure, new standards in innovation, and a closely-knit community of stakeholders

Pharma can be an insular industry, with limited capacity to gaze beyond its own narrow frame of vision

Head of the Publications and Multimedia Department

To manage through a resource-constrained environment, it is essential to get the most out of procurement planning

January 10, 2011.

The mediterranean nation has endeared itself to foreign businesses over the past decades, adtively positioning itself as an appealing epicenter of global pharma interest
The Oxford Finance Programme for Senior Executives is for directors from technical backgrounds involved in strategic planning, and delivers the ability to identify which projects to invest in, retain or divest.

After many years of building up an impressive resource base, the industry is finally ready to take advantage of the wave of enthusiasm and interest surounding pharma and biotech that is sweeping the nation

A small company shows how it can play a major role in a relatively short time frame

Outgoing EMA Director Thomas Lonngren discusses a decade at the helm of Europe's top regulatory office

Cegedim survey documents whether European industry is ready for the challenge of new compliance and disclosure risks on promotional spend

November 6, 2010.

With all the money and momentum focused on medicines in emerging markets, it may be time to take a closer look at business opportunities in this more familiar neighbor to the East

Pharmaceutical Executive
A European pilot initiative will test the impact of consultations with a number of stakeholders such as patient representatives and payers on early stage drug development.

As part of a team, you will prepare meetings of European Pharmacopoeia groups of experts, contribute to the preparation of working documents for these groups, contribute to the elaboration and revision of European Pharmacopoeia texts and follow up meetings with a view to publication of scientific texts and memoranda.

Pharmaceutical Executive
A report by the European Commission aimed at exploring patent exploitation by Big Pharma winds up generating more angst than answers

September 21, 2010.

Pharmaceutical Executive
Anjan Selz offers a Strengths, Weaknesses, Opportunities and Threats analysis of the biosimilars market.

September 08, 2010.

Dr Frank Floether provides a no-nonsense guide to securing a successful alliance in Asia.