
In today’s Pharmaceutical Executive Daily, learn how Abbott is acquiring Exact Sciences in a major diagnostics deal, Novartis is building a new manufacturing hub in North Carolina, and the FDA is rolling out a wide batch of drug approvals.

In today’s Pharmaceutical Executive Daily, learn how Abbott is acquiring Exact Sciences in a major diagnostics deal, Novartis is building a new manufacturing hub in North Carolina, and the FDA is rolling out a wide batch of drug approvals.

Tesaro and AnaptysBio engage in legal battles over alleged breaches of their 2014 partnership agreement regarding Jemperli.

Pharmaceutical collaborations expand as Flagship Pioneering expands its partnership with GSK, signing agreements with ProFound Therapeutics and Quotient Therapeutics, while Valo Health teams up with Merck KGaA to tackle disease challenges.

Jay Bregman, co-founder of Andel, the cost and operational complexity of covering GLP-1 medications.

In today’s Pharmaceutical Executive Daily, learn how Moderna is expanding its manufacturing footprint in Massachusetts, GSK is advancing new research collaborations under the LTZ Fleming Initiative, and the FDA commissioner highlights how early and late movers can benefit from the National Priority Voucher program.

The FDA approves multiple new drugs, enhancing treatment options for cancer and other conditions.

As part of its multi-billion-dollar investment, the company will be creating a unified, end-to-end manufacturing campus spanning biologics, gene therapy, solid dosage, and packaging operations.

Abbott's acquisition of Exact Sciences enhances cancer diagnostics and expands access to innovative screening and treatment solutions for patients worldwide.

In the first part of our conversation with Ryan Conrad, visiting fellow at the Center on Health Policy at the Brookings Institution, he provides a broad overview of the possible positive and negative impacts of FDA’s Priority Review Voucher Program.

In today’s Pharmaceutical Executive Daily, learn how Novo Nordisk is launching an introductory offer for Wegovy and Ozempic, AbbVie and Genmab gain FDA approval for Epkinly in follicular lymphoma, and Johnson & Johnson announces a $3 billion definitive agreement to acquire Halda Therapeutics.

GSK's collaborations leverage cutting-edge technology and scientific expertise to advance and accelerate research.

Moderna invests $140 million in its Norwood facility, creating hundreds of bio jobs while also enhancing manufacturing presence in the U.S.

The first wave of Commissioner’s National Priority Vouchers signals a fundamental shift in FDA competitiveness, rewarding companies that pair breakthrough science with affordability commitments, onshore manufacturing readiness, and the operational muscle to execute ultra-accelerated reviews.

A US-China Economic and Security Review Commission report highlights China’s tightening grip on APIs, biomanufacturing, and R&D services, raising alarms about supply chain vulnerabilities, data transparency gaps, and the urgent need for US policy action.

Avadel Pharmaceuticals accepted Alkermes enhanced acquisition proposal for $2.3 billion, focusing on a key treatment's approval for added value.
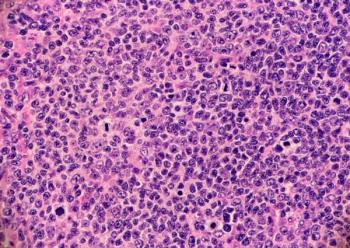
Epkinly plus rituximab and lenalidomide is the first bispecific antibody combination FDA-approved for relapsed or refractory follicular lymphoma, backed by Phase III data showing substantially improved disease control over standard therapy.

Craig Ackerman, partner in the Alexander Group and lead of its medical device practice, discusses how AI is being used and whether pharma companies should brace for the bubble to pop.

Johnson & Johnson aims to enhance its oncology portfolio and innovate prostate cancer treatments by expanding its therapeutic pipeline through the acquisition.

The FDA’s new Commissioner’s National Priority Voucher signals a transformative shift in U.S. drug review, tying accelerated approval to manufacturing readiness, domestic supply-chain strength, and credible affordability commitments, while redefining how companies must innovate, scale, and launch therapies in 2025 and beyond.

In today’s pharmaceutical executive daily learn how Novo Nordisk is restructuring leadership to enhance scientific and commercial alignment, ABL Bio secures a major multi-program partnership with Eli Lilly, and the FDA launches its new Plausible Mechanism Pathway to accelerate drug development.

UnitedHealth Group appoints Dr. Scott Gottlieb to its board, enhancing leadership amongst a time in which the company looks to enhance its public outlook.
The company is launching new self-pay pricing options, expanded savings programs, and partnerships with telehealth and retail providers. all while its obesity portfolio gains momentum under the FDA’s National Priority Voucher program.

Eli Lilly partners with ABL Bio to research innovative therapeutics and expand its portfolio in cancer and CNS diseases through advanced bispecific antibody technology.

Start managing the transformation now to secure every benefit.

Jay Lalezari, CEO, CytoDyn, emphasizes how regulatory T cells play a crucial role in tumor progression by enabling cancers to evade immune detection.

Novo Nordisk’s board overhaul signals a decisive return to Foundation-led scientific and operational governance, raising strategic questions about whether the company can sustain U.S. growth and consumer-market leadership in the GLP-1 era without deeper American commercial expertise.

Olympia Pharmaceutical’s CEO discusses the company’s expansion into Mississippi.

A Q&A with Christopher Stanton, Ph.D., Marvin Bower Associate Professor of Business Administration at Harvard Business School.

Dr. Jaspal Singh explains some of the barriers that prevent cancer patients from getting biomarker testing early in their treatment journey.

Craig Ackerman, partner in the Alexander Group and lead of its medical device practice, explains the differences in strategy based on which region you’re strategizing for.