
In today’s Pharmaceutical Executive Daily, we cover Glenmark’s billion-dollar exclusive licensing deal with Hengrui, Harmony Biosciences’ trial setback for Zygel, and the CDC’s unanimous vote to broaden COVID-19 vaccine recommendations.

In today’s Pharmaceutical Executive Daily, we cover Glenmark’s billion-dollar exclusive licensing deal with Hengrui, Harmony Biosciences’ trial setback for Zygel, and the CDC’s unanimous vote to broaden COVID-19 vaccine recommendations.
Ascletis Pharma reveals ASC47, a promising weight loss drug, shows over 56% greater body weight reduction when combined with semaglutide in obesity treatment.

The company did not provide a reason for the bimagrum trial’s halting.

Pharma leaders embrace direct-to-consumer strategies, with 94% exploring DTC programs to enhance patient experience and accelerate treatment access.
The arrival of GLP-1 receptor agonists and dual agonists has delivered unprecedented efficacy, reframed obesity as a chronic disease, and sparked an industry gold rush.

In today’s Pharmaceutical Executive Daily, we cover the market reaction to Trump’s Tylenol–autism claim, the FDA’s complete response letter to Scholar Rock for its spinal muscular atrophy treatment, and a closer look at the state of evidence linking Tylenol use and autism risk.

CDC vaccine advisors shift to shared decision-making for Covid-19 vaccines, emphasizing patient-provider consultations amid insurance concerns.
Following the President’s claims connecting the drug to an increased risk of autism when used during pregnancy, Tylenol has found itself in an unexpected spotlight.

Harmony Biosciences reveals disappointing results from the Phase III Reconnect study of ZYN002 for Fragile X Syndrome, highlighting ongoing research challenges.

Glenmark partners with Hengrui Pharma to develop Trastuzumab Rezetecan, enhancing its oncology pipeline and addressing unmet cancer treatment needs.

In today’s Pharmaceutical Executive Daily, we cover GSK’s supplemental application to update the label for Leucovorin, the FDA’s complete response letter to Otsuka and Lundbeck’s sNDA for Rexulti, and the medical community’s response to President Trump’s claims linking Tylenol use to autism risk.

Investor reaction is showing how the industry is reacting to the President’s claims.

GSK seeks FDA approval for Leucovorin to treat cerebral folate deficiency and autism symptoms, promising new hope for affected children.

Scholar Rock navigates FDA's Complete Response Letter for Apitegromab, aiming for swift resubmission to address spinal muscular atrophy treatment needs.

The President announced FDA will no longer recommend the use of the common painkiller during pregnancy, despite a casual link between the medication and autism not being established.

In today’s Pharmaceutical Executive Daily, we cover Biogen’s acquisition of Alcyone Therapeutics to expand its drug delivery solutions, the FDA’s expanded approval of Incyte’s Opzelura for atopic dermatitis in young children, and Pfizer’s move to acquire Metsera to strengthen its obesity and cardiovascular pipeline.

FDA denies approval for Rexulti in PTSD treatment, citing insufficient evidence despite clinical trials. Otsuka and Lundbeck plan next steps.

Pfizer acquires Metsera for $4.9 billion, enhancing its portfolio in obesity and cardiovascular treatments with innovative therapies and strong clinical potential.

In today’s Pharmaceutical Executive Daily, we cover how pharma companies can scale AI pilots into enterprise-wide value, GSK’s plan to invest $30 billion in U.S. biopharma capacity, and Novo Nordisk’s clinical trial success with oral Wegovy.
CDC vaccine advisers delay Hepatitis B schedule changes while recommending separate vaccines for measles, mumps, rubella, and chickenpox amid safety concerns.

Biogen expands its drug delivery solutions by acquiring Alcyone Therapeutics, enhancing patient-centered therapies with the innovative ThecaFlex DRx system.

VectorY Therapeutics partners with Shape Therapeutics to advance innovative therapies for neurodegenerative diseases using SHP-DB1 technology and AAV5 capsids.
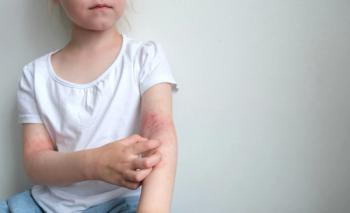
The FDA has expanded the indication of Incyte’s Opzelura (ruxolitinib) cream to include children aged two to 11 years with mild to moderate atopic dermatitis, making it the first steroid-free topical treatment option for this age group.

In today’s Pharmaceutical Executive Daily, we explore how AI is disrupting higher education and career pathways, AllRock Bio’s $50 million Series A funding to advance its lead molecule, and FDA warning letters issued to Novo Nordisk, Eli Lilly, and Hims over misleading drug advertising.
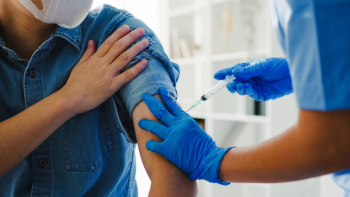
Former CDC director Susan Monarez raises alarms about potential changes to childhood vaccine schedules, warning of increased health risks for children.

Novo Nordisk's shares rise as Phase III trial shows its oral Wegovy pill achieves significant weight loss, offering a new option for patients.

The company announced the plans as President visited the UK.

Roche strengthens its pipeline by acquiring 89bio and its promising MASH treatment, Pegozafermin, enhancing options for metabolic disease therapies.

In today’s Pharmaceutical Executive Daily, we cover Lilly’s orforglipron as a prime candidate for FDA fast-track approval, Merck and Daiichi Sankyo’s breakthrough therapy designation for R-DXd in ovarian cancer, and Novo Nordisk’s EU approval of Rybelsus for reducing cardiovascular risk.

FDA warns major pharmaceutical companies to improve transparency in drug advertising, addressing misleading claims and enforcing stricter regulations.