Sponsored
Elyse Blazevich discusses the Colorado Bioscience Association’s efforts to support the state’s life sciences sector by fostering partnerships, advocating for industry growth, and building a diverse talent pipeline.

Sponsored
Elyse Blazevich discusses the Colorado Bioscience Association’s efforts to support the state’s life sciences sector by fostering partnerships, advocating for industry growth, and building a diverse talent pipeline.

Sponsored
In this interview, Melissa Krebs,Ph.D., CEO and founder of GelSana, shares her journey in developing cutting-edge biomaterials that could transform the $78 billion wound care market.
Sponsored
Dr Peter Wahl, MLA, MS, Global Head of Scientific Affairs at CorEvitas, part of the PPD clinical research business of Thermo Fisher Scientific, covers key considerations for choosing registries.
Sponsored Video
How a multipronged approach to staff recruitment can accelerate trial success.
Insights into current practice of global pharma labeling, including shortcomings and potential improvements.

While clinical registries are a valued source of longitudinal, real-world data, registries also provide expanded options for research and advancing randomized clinical trials. Understanding when and how to use registries in these use cases is quite nuanced and mandates a targeted approach and innovative expertise. In this video, we dive deep into the expanded use of registries as external control arms in clinical trials.

Pharmaceutical Executive, in collaboration with Biofourmis, organized a roundtable of experts from various groups to discuss how digital medicine and pharmaceuticals are changing the trajectory of what drug development and clinical care look like together, with an emphasis on partnerships within digital health.

Richard Young, Vice President of Strategy, Vault CDMS and Tim Davis, Vice President of Strategy, MyVeeva for Patients, discuss the future of digital trials and their impact in the industry.
Imaging is an integral part of many clinical studies, particularly in the fields of oncology and ophthalmology. Learn why imaging and centralized review are essential for clinical trials, how to address challenges that arise in these studies, and the advantages of partnering with MERIT in this video.

Defining what Gross-to-Net mean and why it is important to Life Sciences companies; Identify who in the company benefits in utilizing information from GTN; Get insights to successfully define and manage GTN process

In this video Akeel Williams, VP of Strategy and Operations, Manufacturer Solutions, explains how GoodRx uses data to continuously improve its platform for patients and providers.
As life science organizations continue to leverage both real world data [RWD] and real-world evidence [RWE] in the product approval landscape, a more nuanced understanding of both RWD and RWE is needed. Given the expansive types of data available now, understanding which data set is needed and how it should be analyzed is decidedly valuable. Our expert provides basic tenets for consideration

Brian Torres of Herspiegel Consulting explains the recent shift in patient support programs and how that will influence the future of Pharma.
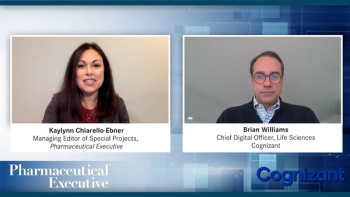
At a time when life sciences companies have seen an unprecedented acceleration in the adoption of digital tools, many are now faced with managing a myriad of tools and applications with varying levels of security and technology requirements. Biopharma and medical device manufacturers are now exploring solutions and platforms that can provide the stability, regulatory compliance, and end-to-end connectivity needed to design, deliver and scale digital solutions that deliver clinical and economic benefits to patients, providers and payers. In this session, Brian Williams, Chief Digital Officer for Life Sciences at Cognizant, will discuss the opportunity available to digital-first companies that are considering cloud platforms like Philips HealthSuite, which is built on Amazon Web Services.

This video series discusses the impact of implementing risk-based quality management (RBQM) and decentralized clinical trials (DCTs) from the clinical operations point of view.
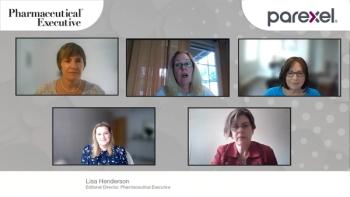
As the initial COVID-19 pandemic begins to resolve in the form of vaccines and treatment pathways, research into the long-term effects of COVID is building. In response, in December, Congress provided $1.15 billion in funding over four years for NIH to support research into the prolonged health consequences of SARS-CoV-2 infection. This roundtable of experts from various stakeholder groups will discuss the current research into long-haul COVID, or PASC; disparate theories of causes; challenges in research and diagnosis, as well as PASC potential in drug development.

Sponsored Videos
In a recent virtual roundtable discussion, industry experts Jim Streeter, Alison Holland, Hassan Kadhim, Craig Lipset, Josh Rose, and Craig Serra shared their insight on the challenges faced in implementing decentralized trials and how to address them. Along with the roundtable are video discussions covering the cost and feasibility of a toolkit approach to decentralized trials, the significance of minimizing burden to patients and sites, and the reality of regulatory hurdles.