Regulatory
Latest News
Latest Videos

More News
The approval is based on Phase III DESTINY-Breast09 data showing the Enhertu–Perjeta combination significantly improved progression-free survival compared with the current taxane-based standard of care in previously untreated metastatic disease.
The FDA’s approval of Lerochol introduces a once-monthly, subcutaneous PCSK9 inhibitor for lowering LDL cholesterol in adults with hypercholesterolemia.

The 16th addition to the FDA Commissioner’s National Priority Voucher pilot program follows Phase III MajesTEC-3 trial data showing significant survival benefits for the Tecvayli-based regimen compared with standard of care in treating relapsed or refractory multiple myeloma.

FDA Approves Amgen's Uplizna for Generalized Myasthenia Gravis With Positive AChR or MuSK Antibodies
The FDA’s approval of Uplizna for antibody-positive generalized myasthenia gravis introduces a twice-yearly CD19-targeted therapy option for the rare autoimmune condition.

TrumpRx’s promised drug-price cuts expose a deeper systemic flaw, positioning direct-to-patient clinical pathways as the infrastructure pharma must build to turn political headlines into real access and outcomes.

In the third and final part of her conversation with Pharmaceutical Executive, discusses the ways that regulatory agencies in major global markets are acting in similar ways to have a positive impact on the biosimilars market.

Experts weigh in on FDA’s new accelerated review program, revealing what companies stand to benefit the most from the initiative.

Amid the US reshoring push, drugmakers navigate geopolitical and supply chain risks.

In the second part of her conversation with Pharmaceutical Executive, Gillian Woollett, VP and head of regulatory strategy at Samsung Bioepis, explains the direct impact changes at FDA have on the biosimilar space.

FDA’s approval of Augmentin XR as the first therapy cleared under the new National Priority Voucher Pilot Program signals a decisive push to strengthen domestic antibiotic manufacturing, accelerate critical drug reviews, and stabilize long-strained U.S. antimicrobial supply chains.

New FDA guidance signals a major shift for CAR-T development, calling for randomized trials with standard-of-care control groups and clear evidence of superiority over existing therapies, while simultaneously easing REMS requirements to reduce logistical burdens for treatment centers and patients.

Welcome to Pharm Exec's new guest column on navigating the geopolitical risks challenging core company strategies in today's shifting global landscape for healthcare.

Gillian Woollett, VP and head of regulatory strategy at Samsung Bioepis, discusses how the current regulatory climate is the result of years of work.

FDA’s approval of Bristol Myers Squibb’s Breyanzi as the first CAR T therapy for relapsed or refractory marginal zone lymphoma underscores the therapy’s expanding leadership in B-cell malignancies, backed by compelling Phase II data

Former FDA Commissioners Warn New Vaccine Policies Could Undermine Longstanding Regulatory Framework
Twelve former FDA leaders have publicly challenged the agency’s proposed overhaul of vaccine approvals, sparked by an internal memo linking child deaths to COVID-19 vaccination, arguing the changes threaten evidence-based standards, weaken immunobridging practices, and risk eroding public trust.

The agency based its approval off results collected from the Bruin CLL-321 trial.

The agreement includes exemptions for UK-produced drugs and medical devices from Section 232 tariffs, but mandates a significant change to the UK's NICE value appraisal framework.
The agency’s decision is based on claims that the Covid vaccine was linked to the deaths of 10 children.

In the final part of this conversation, Dr. Bruce Leuchter, CEO of Neurvati Neurosciences, discusses new programs at FDA and how the neuroscience space is benefitting.

The company submitted the new dosage for approval based on its Phase III trial results.

In the first part of our conversation with Ryan Conrad, visiting fellow at the Center on Health Policy at the Brookings Institution, he provides a broad overview of the possible positive and negative impacts of FDA’s Priority Review Voucher Program.

The first wave of Commissioner’s National Priority Vouchers signals a fundamental shift in FDA competitiveness, rewarding companies that pair breakthrough science with affordability commitments, onshore manufacturing readiness, and the operational muscle to execute ultra-accelerated reviews.

A US-China Economic and Security Review Commission report highlights China’s tightening grip on APIs, biomanufacturing, and R&D services, raising alarms about supply chain vulnerabilities, data transparency gaps, and the urgent need for US policy action.
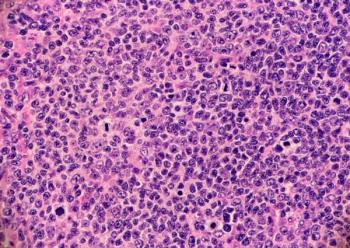
Epkinly plus rituximab and lenalidomide is the first bispecific antibody combination FDA-approved for relapsed or refractory follicular lymphoma, backed by Phase III data showing substantially improved disease control over standard therapy.

The FDA’s new Commissioner’s National Priority Voucher signals a transformative shift in U.S. drug review, tying accelerated approval to manufacturing readiness, domestic supply-chain strength, and credible affordability commitments, while redefining how companies must innovate, scale, and launch therapies in 2025 and beyond.