
Pharmaceutical Executive
Although pricey, serialization with RFID is expected to reduce logistical errors and address some aspects of supply chain security

Pharmaceutical Executive
Although pricey, serialization with RFID is expected to reduce logistical errors and address some aspects of supply chain security

Pharmaceutical Executive
With gaming technology, pharmaceutical companies can display immersive 3-D animation that allows doctors to explore inside an interactive environment pertaining to a drug's method of action

Pharmaceutical Executive
Drug giant moves corporate, promotional communications online.

Pharmaceutical Executive
Product sites need to offer consumers more than just a hard sell.

Pharmaceutical Executive
Documents from R&D, clinical affairs, regulatory, and sales and marketing can be in the millions. Throw electronic information into the mix, and the number of documents required for litigation increases exponentially.

Pharmaceutical Executive
Doctors like direct mail as long as the message is concise and to the point. The minute you ask the doctor to actually do something with the direct mail, that's when your effectiveness starts to go downhill.

Pharmaceutical Executive
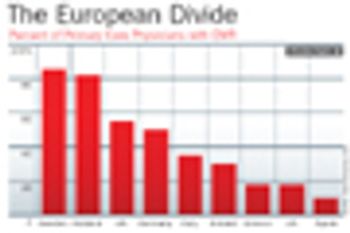
After decades of disparate attempts to secure supply chains, pharma companies may finally be getting on the same page.
Pharmaceutical Executive
Clinical researchers cannot reliably use many of today's electronic health records because of the variability among collection systems.

Pharmaceutical Executive
New partnerships with nanobiotech firms are helping pharma companies overcome solubility problems and extend profitable product lifecycles.
Pharmaceutical Executive
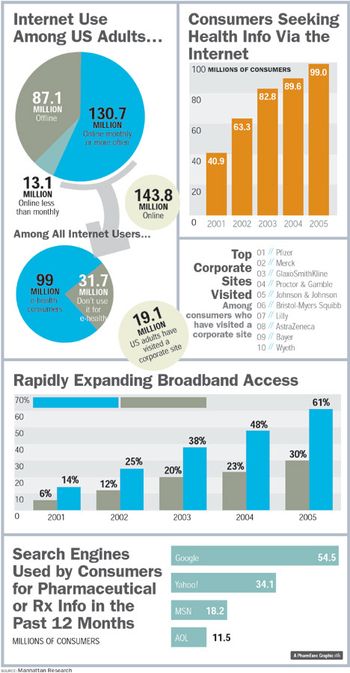
When people ask how health seekers look and act on the Web, there's no one answer: The online universe has become just as diverse as the rest of the world. Nowhere is this more evident than in the latest e-health statistics that, when taken together, paint a picture of customers who are beginning to find their feet-and new finesse-online.

Pharmaceutical Executive
Jeffrey Kindler holds two blue, diamond-shaped pills in the palm of his hand. One is authentic Viagra, manufactured by Pfizer. The other is counterfeit, maybe bought by an undercover Pfizer investigator, or intercepted when smugglers crossed a border, or perhaps seized in a raid on an illegal Chinese factory. Kindler challenges visitors and fellow employees to tell the difference between the two pills. Neither looks in any obvious way "fake," and no one among the journalists, corporate communications employees, or even security specialists gathered in Pfizer's global security operations center cares to hazard a guess.

Pharmaceutical Executive
Adaptive trials aren't just for propeller-heads anymore. They're one of the issues that need to be top-of-mind for the whole executive suite, as a driver of new processes and timelines, as a hot-spot on the budget, and as a battleground where public policy on drug safety and efficacy will be fought out.

Pharmaceutical Executive
I needed to eat. I even wanted to eat. Instead, I sat in front of my hospital lunch tray, unable to face up to a carton of red Jell-O. When you think of it, that's a situation most of us face (minus the Jell-O) every day in business.

Pharmaceutical Executive
Electronic data capture (EDC) is an emerging paradigm for gathering information in clinical trials. Ask anyone who has sorted through stacks of accumulated paper at the end of a study, and they'll say EDC is the wave of the future. But while many companies are on board with the technological benefits, enthusiasm wanes when it comes to actual implementation. Even some of EDC's biggest champions admit to its obstacles: "It's a disruptive technology that doesn't give immediate returns," says James Tiede, vice president of integrated data services, global clinical operations at Johnson & Johnson.

Pharmaceutical Executive
RFID is not ready for prime time anywhere. Certainly not in the US. There is no way RFID gives you end-to-end control of the product.

Pharmaceutical Executive
In a product recall, manufacturers would know which retailers and hospitals got bad bottles. Instead of clearing shelves everywhere, they could call specific customers and say, "Hey, we know you have five of these bottles. We want them back."

Pharmaceutical Executive
Scott Gottlieb called RFID implementation "disappointing." Now, FDA is moderating its view of RFID as a cure-all for the industry.

Pharmaceutical Executive
The Zelnorm study was conducted for $1,000 per patient, a fraction of the price of clinical trials, which can cost $10,000 per patient or more.

Pharmaceutical Executive
Online strategies aren't monolithic; each must meet the particular goals of the brand in question, and serve the knowledge needs of a specific disease state. That said, experts agree on many of the fundamentals-the 5 "I"s of Internet marketing:

Pharmaceutical Executive
At least five of the world's top pharma companies have specified, tested, or deployed large-scale Wi-Fi programs.

Pharmaceutical Executive
The most important result for pharma firms of the changing sales structure is a reduction in the amount of training resources that are available to them.

Pharmaceutical Executive
When you have a workable technology, the question becomes ‘To what do you apply this technology?’ and ‘Where do you spend your time?��’ We really believed we would be most successful by spending a lot of time figuring out which drugs to work on and then working assiduously on those few products with huge potential.

Pharmaceutical Executive
Digital forms of image transmission, archiving, and education with the virtual microscope will revolutionize pathology.

Pharmaceutical Executive
The issue of pedigree requirements is emerging at the state level-and figuring out to what extent that will push back to the manufacturers.

Pharmaceutical Executive
Imagine drugs that can detect one particular compound in a patient's body and respond to it by releasing a drug. They're not that far away.