
Pharmaceutical Executive
Pharma companies believe that they can compete in an e-prescribing environment if information systems permit full disclosure and allow doctors to create bookmarks that link easily to information they want.

Pharmaceutical Executive
Pharma companies believe that they can compete in an e-prescribing environment if information systems permit full disclosure and allow doctors to create bookmarks that link easily to information they want.

Pharmaceutical Executive
Without common reporting standards in place, researchers have little incentive to share data with scientists elsewhere in the company. When researchers don't sharedata on a regular basis, they can begin to feel proprietary about their work-and even less inclined to disclose their results.

Pharmaceutical Executive
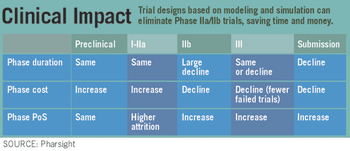
Pharma companies are committed to using electronic data capture in clinical trials. Technology adoption will continue to grow, as FDA and consumers want faster safety data.

Pharmaceutical Executive
CMS envisions studies to show which drugs keep patients out of hospitals or how certain treatments can reduce side effects. Such analysis would support decisions on best practices in using medications.

Pharmaceutical Executive
Even though data can single out physicians with high marketing upsides, most pharma companies are doing without such high-value data.

Pharmaceutical Executive
The future of US healthcare is being created today in Medicare's demonstration programs. But how you respond to them depends a lot on what kind of company you are.

Pharmaceutical Executive
In a non-human primate study, high doses of Neumune resulted in 90 percent survival against a lethal level of radiation.
Pharmaceutical Executive
The use of a CUI leads development efforts to explre the most valuable region of treatment - not necessarily the the most efficacious.

Pharmaceutical Executive
In an ideal world, an anti-counterfeit solution would provide protection throughout the supply chain, allow for easy product identification by physicians, pharmacists, and patients, be easily implemented without ongoing costs-and improve brand image and marketability while it's at it. Yet most current anti-counterfeiting measures involve packaging technologies such as holograms, inks, bar codes and radio frequency ID (RFID) that, although useful, cannot ensure the integrity of the pharmaceutical supply chain, because drugs do not remain in their original packaging. Legitimate repackaging regularly occurs in the pharmacy and elsewhere, and authentic packaging-recycled or stolen-can contain adulterated, counterfeited drugs.