
The use of artificial intelligence (AI) in drug development has been hampered by issues that have caused a gap between AI’s potential and its full utilization in this space.

The use of artificial intelligence (AI) in drug development has been hampered by issues that have caused a gap between AI’s potential and its full utilization in this space.
Joint venture aims to submit an Investigational New Drug application to the FDA by early next year for treatment that targets solid tumors.

Novel oral orexin receptor 2 agonist produced statistically significant and clinically meaningful improvements in wakefulness compared with placebo in patients with narcolepsy type 1.

In an interview with Pharm Exec Associate Editor Don Tracy, Jonathan Miller, General Counsel, US Hemp Roundtable, discusses recent efforts that urge the House Energy and Commerce Committee to expedite an FDA hearing on the hemp market.

Researchers evaluate a potential association between the use of phosphodiesterase type 5 inhibitors for erectile dysfunction and a reduced risk of Alzheimer disease.
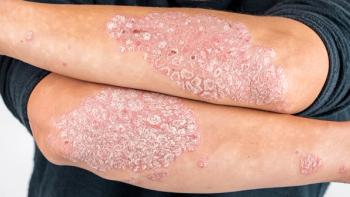
JNJ-2113, an oral IL-23–receptor antagonist peptide, showed consistency across clinician and patient-reported outcomes in patients with moderate-to-severe plaque psoriasis.

Data from the ENHANCE-3 trial of magrolimab in combination with azacitidine plus Venclexta showed futility and an increased risk of death in patients with acute myeloid leukemia.

As the market for biosimilars expands, a nuanced approach is needed to balance cost considerations with patient safety and pharmacovigilance efforts.
The biopharma company’s Phase 3 trial for its acoramidis product generated statistically significant results.

The Maryland facility will create cell therapies for use in future cancer trials.

Results from the Phase III CheckMate -77T trial show that Opdivo (nivolumab) plus chemotherapy followed by surgery and adjuvant Opdivo produced statistically significant and clinically meaningful improvements in event-free survival in patients with resectable stage IIA to IIIB non-small cell lung cancer.
Vepdegestrant (ARV-471) is a noval PROTAC ER degrader found to harness the body’s own natural protein disposal system to eliminate disease-causing proteins.

The agreement will strengthen Novartis’ oncology pipeline.
Cancer & Work: Acting Together guarantees job security and full salary continuation for up to twelve months for Sanofi employees and family members diagnosed with critical illnesses.

In a Phase I trial, data suggest that Amgen's maridebart cafraglutide (MariTide) may allow patients to take lower and less frequent doses over time while still maintaining significant weight loss.
Patients with relapsed or refractory multiple myeloma administered Blenrep combined with bortezomib plus dexamethasone experienced a 59% reduction in the risk of disease progression or death compared with the standard of care.
The report predicts a 77% increase in global cancer cases by 2050.
BioNTech SE and Duality Biologics' next-generation antibody-drug conjugate is being evaluated for patients with platinum-resistant ovarian epithelial cancer, fallopian tube cancer, or primary peritoneal cancer previously administered one to three systemic treatment regimens.
The FDA assigned a Prescription Drug User Fee Act action date of June 7, 2024, to an application that would expand the indication of Arexvy to include adults 50-59 years with an increased risk of respiratory syncytial virus-related lower respiratory tract disease.

Synchron’s brain-computer interface device is being developed to allow patients with mobility challenges to operate certain technology with their mind.
UV1 plus ipilimumab (Yervoy) and nivolumab (Opdivo) produced a statistically significant and clinically meaningful survival improvement in patients with unresectable malignant pleural mesothelioma.
The $1.3 billion acquisition—which included two aqua manufacturing plants—expands Merck’s portfolio in the veterinary pharma space.

By capitalizing on AI advancements, pushing the boundaries of personalized medicine with theranostics, and strengthening digital healthcare networks, radiopharmaceuticals are poised to transform healthcare from a reactive to a proactive discipline.
Higher prices of insulin attributed to significant annual increases, outpacing general inflation.

The era of big pharma as product-first companies must end, as services become the larger priority.
The drugmaker will directly acquire three finish-fill sites as part of the deal.

In an interview with Pharm Exec Associate Editor Don Tracy, Shubh Goel, Head of Immuno-Oncology and Gastrointestinal Tumors, US Oncology Business Unit, AstraZeneca, provides her thoughts on the success of the trial.
Beyfortus (nirsevimab-alip), a monoclonal antibody that protects against respiratory syncytial virus-associated lower respiratory tract disease, experienced higher than anticipated demand that led to shortages during the 2023-2024 season.

The agency issued the warning against three brands of unapproved eyedrops designed to look like an approved brand.

Novartis rescinds rights to to develop and commercialize multi-tyrosine kinase inhibitor dovitinib due to a material breach by Allarity for lack of financial payment.