The biologics license application for afamitresgene autoleucel, an engineered T-cell receptor drug, was assigned a PDUFA date of August 4, 2024.

The biologics license application for afamitresgene autoleucel, an engineered T-cell receptor drug, was assigned a PDUFA date of August 4, 2024.
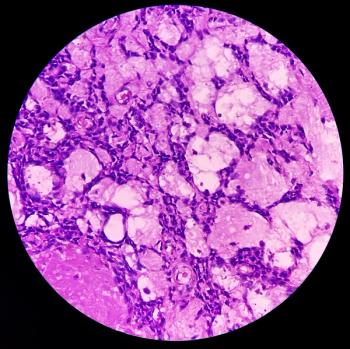
BST02 is the first tumor-infiltrating lymphocyte therapy for the treatment of all types of liver cancer to have advanced to the clinical trial stage.
The US Department of Health and Human Services has initiated a comprehensive strategy to address the escalating syphilis rates, as a result.

DELFI-Tumor Fraction assay was developed to improve noninvasive assessment of tumor burden and monitoring of treatment efficacy and resistance in patients with advanced cancers.
Rusfertide is currently in a pivotal Phase III clinical trial as a potential first-in-class treatment for polycythemia vera.
Vabysmo is the first bispecific antibody approved to treat ocular conditions such as diabetic macular edema and wet age-related macular degeneration.

App aims to leverage digital technology to significantly reduce pharmacy costs.
Aduhelm (aducanumab-avwa) was granted accelerated approval by the FDA in June 2021 despite misgivings from the agency's Peripheral and Central Nervous System Drugs Advisory Committee.

In an interview with Pharm Exec associate editor Don Tracy, Lynn Kirkpatrick, PhD, CEO, Ensysce Biosciences, discusses the FDA's Breakthrough Therapy Designation for PF614-MPAR, which focuses on protecting patients from drug overdoses.
Darzalex Faspro has previously been approved by the FDA for eight indications in multiple myeloma.
Approval sought for Padcev (enfortumab vedotin) plus Keytruda (pembrolizumab) for the first-line treatment of adults with previously untreated locally advanced or metastatic urothelial cancer, which would be the first approved combination alternative to platinum-containing chemotherapy for this patient population.

Eli Lilly advertisement emphasizes that advancements in in medicine can lead to improved population health.

Researchers noted the onset of symptoms in patients who were treated with contaminated HGH therapy.

CAR T-cell therapy Breyanzi (lisocabtagene maraleucel) to be evaluated in patients with relapsed or refractory follicular lymphoma and mantle cell lymphoma following treatment with a Bruton tyrosine kinase inhibitor.

This is Takeda's second approval in chronic inflammatory demyelinating polyneuropathy this month after the FDA approved HyQvia to protect against relapse of neuromuscular disability and impairment in this patient population.

SRP-5051 administered every four weeks produced higher increases in dystrophin and exon skipping compared to eteplirsen dosed weekly in patients with Duchenne muscular dystrophy amenable to exon 51 skipping.
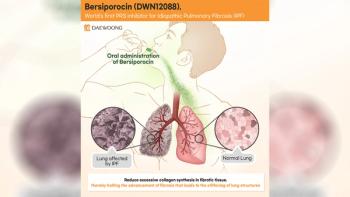
The agency recognized the medication as a significant advancement for the treatment of IPF.

JAMA study evaluates a potential correlation between new recreational cannabis laws and a possible reduction in synthetic opioid deaths.
More than 1600 trials are currently evaluating Keytruda across a range of cancer types and treatment settings.
Enhertu has been approved by the FDA for indications in breast cancer, non-small cell lung cancer, and gastroesophageal junction adenocarcinoma.

In an interview with Pharm Exec Associate Editor Don Tracy, Ron Lanton, Partner, Lanton Law PLLC, discusses recent moves by CVS and Express Scripts to follow Mark Cuban's Cost Plus Drugs model.

Atypical antidepressant linked to severe adverse effects and death as a result of misuse.
Study finds that individuals who took GLP-1 agonists for a long duration had a lower risk of going on to develop more severe forms of liver disease, including cirrhosis and liver cancer.

Substance use disorder is a complex condition that demands innovative solutions, one of which that has shown promise is the use of long-acting injectables.

Report comes after the FDA seized thousands of counterfeit Ozempic units.

The FDA previously approved Dupixent in May 2022 to treat eosinophilic esophagitis in patients aged 12 years and older.

FDA leadership notes that the overall rate of secondary T-cell cancers among patients administered CAR T-cell therapies appears to be low, even if all reported cases are assumed to be related to treatment.

The expanded indication for Zynrelef now includes soft tissue and orthopedic surgical procedures.
Theratechnologies said it will address the FDA’s complete response letter and will continue to seek approval for the new formulation of tesamorelin for the treatment of lipodystrophy in patients also diagnosed with HIV.

Severe hypocalcemia was found to be more common among patients with advanced chronic kidney disease with mineral and bone disorder who are taking Prolia for osteoporosis.