
Companies that want to tap into the full potential of Asia-Pac must boost the role of their regional headquarters.

Companies that want to tap into the full potential of Asia-Pac must boost the role of their regional headquarters.

EU and UK industry groups did their best to display calm and confidence as the world woke up to the reality of Brexit.

Pharmaceutical Executive
A new industry working group explores alternative drug funding options in Europe-and asks key stakeholders in the region to join the discussion.

Do recent trends in the pharma and biotech M&A and equity markets signal a temporary lull - or a permanent drop? Peter Young reports.

Radicals who wield real power at the EMA have gone public with their thinking about the role of regulators in the economic aspects of medicines provision – despite the decades-old EU taboo about linking pricing with authorizations.

NHS England head Simon Stevens believes the UK's PPRS needs significantly updating.

Pharmaceutical Executive
While the Italian fashion and automotive industries may steal the global thunder, another sector in Italy flourishes beneath the public’s radar. The country’s pharmaceutical industry continues to punche above its weight in many areas and has supported Italy’s national economy through many periods of weakness.

We desperately need new anti-infective drugs to avoid life threatening infections. Let’s make that happen by punishing companies who don’t invest in R&D for these specific drugs.

Is a more "light-touch" appraisal process the key to getting the UK's NICE to do more with less? Leela Barham reports.

The dilemma continues: Which medicines should Europe's health systems pay for – and how much should they pay?

As companies look to raise capital, selling and then leasing back real estate is a tempting option and one that other industries are utilizing, writes Karen Williamson.

Pharmaceutical Executive
while France remains a global force in the pharmaceutical, medical device, and vaccines fields-and continues to tout a solid manufacturing base and re-exportation culture-questions are arising over the long- term sustainability of the country’s healthcare system and its traditionally generous public health provisions.

Pharmaceutical Executive
Egypt is not just a sun-soaked paradise defined by pyramids and tombs, or-alternatively-a hotbed for political turmoil and revolution. It’s also a vibrant and growing market for pharmaceuticals, experts contend.

Deep in the massive bureaucracy of the United Nations is an undertaking that appears to have been conceived and designed to be combative - and biopharma may be the target. Stephen Tepp reports.


NICE's notion to charge the industry for its technology appraisals looks set to become reality, writes Leela Barham.

Pharmaceutical Executive
A new wave of technologies supported by innovative business models is transforming the vaccine landscape - and raising the bar on performance. As the demand for cures for chronic diseases accelerates, and with more global outbreaks of viral diseases like Zika and Ebola a virtual certainty, solutions can’t come soon enough.

Pharmaceutical Executive
A new study spells out the many drug pricing problems in Europe just fine, but fails to address a central access issue-leaving its recommendations frustratingly limited for industry and policymakers.

Bruno Villetelle discusses how Japanese companies like Takeda have begun to digitize in order to care for the country's aging population.

Pharmaceutical Executive
The Dutch healthcare system, dubbed today as a “laboratory for change,” is in the midst of reinventing itself around collaborations with industry and government in such areas as market and patient access, transparency, cost-effectiveness, and process innovation.

As a European politician attacks abuses of the orphan drug scheme, a European report extols its virtues. Reflector asks: in Europe, does the right hand know what the left hand is doing?
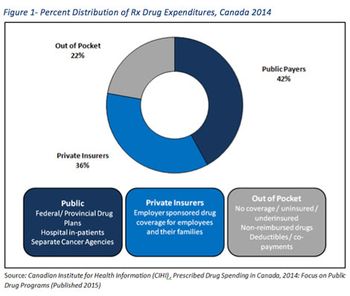
Arvind Mani and Sherry O' Quinn outline the prevalence of private payer PLAs in Canada.

Following the consultation on the future of England's Cancer Drug Fund, Leela Barham reviews what the stakeholders are thinking.

Pharmaceutical Executive
Whatever used to be wrong with the world of big Pharma could be fixed with a single tag phrase: emerging country markets. Most of the majors have invested heavily in this geographic segment, and the biggest of the big-companies like Novartis-now rely on it for more than a quarter of their global sales. Like all good things, however, there are shadows amidst the sunlight, and the task of turning volume sales into sustainable profits is getting harder.

Pharmaceutical Executive
Pharm Exec interviews Subhanu Saxena, CEO of India pharma giant Cipla, who discusses the longtime emerging market champion’s strategic plans to raise its geographic profile and secure the company’s future in the US.