Superluminal partners with Eli Lilly to develop AI-driven small molecule therapies targeting GPCRs for obesity and cardiometabolic diseases, aiming for innovative treatments.

Superluminal partners with Eli Lilly to develop AI-driven small molecule therapies targeting GPCRs for obesity and cardiometabolic diseases, aiming for innovative treatments.

Rob Abbott, CEO, ISPOR, highlights how emerging data strategies, advanced technology, and a renewed focus on patient needs are setting the direction for the next era of healthcare worldwide.

The federally created panel was previously disbanded in 1998.

Rob Abbott, CEO, ISPOR, explains how health economics and outcomes research is helping healthcare systems measure the true value of treatments, improve patient outcomes, and address rising drug costs globally.

The milestone payment is partially funded through Sarepta’s Arrowhead stock sale as both companies push forward with promising RNA interference programs.

The directive calls for building a Strategic Active Pharmaceutical Ingredients Reserve, updating the essential medicines list, and prioritizing US-made APIs.

Fate Therapeutics, Generation Bio, and ORIC Pharmaceuticals join a growing list of companies implementing workforce reductions as they restructure operations, extend cash runways, and focus resources on advancing lead clinical programs amid challenging market conditions.
Connect Biopharma partners with the Jovante Woods Foundation to enhance asthma education and awareness, aiming to prevent acute attacks and save lives.
The new list price will bring the medication in line with other European markets.

Seal Rock Therapeutics joins The Michael J. Fox Foundation's LITE program to help advance research and developments in inhibitors targeting LRRK2 in Parkinsons disease.
Oikos Fusion offers a nutrient-dense dairy drink designed to support muscle mass and digestive health during weight loss journeys.

Users find themselves suddenly looking for an extra $500-a-month in their budgets.

The collaboration will fund early-stage clinical development of SB-4826, CIT’s first-in-class SUMO E1 inhibitor, for patients with relapsed or refractory follicular lymphoma.

The project aims to enhance AbbVie’s chemical synthesis capacity to support next-generation neuroscience, immunology, and oncology medicines, with construction beginning in 2025 and operations slated for 2027.

Attorney General Paxton argues that the company’s programs and offerings were essentially bribes and resulted in millions in dollars charged to the state’s Medicaid program.
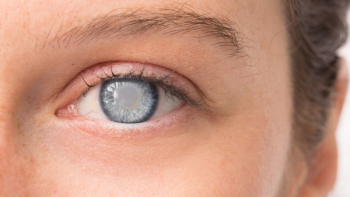
Monica Seles partners with argenx to raise awareness for Myasthenia Gravis, sharing her journey to empower and support those affected by the condition.

The specialty medication platform earned a spot in the top 15% of the list.

Bayer partners with Kumquat Biosciences to advance a promising Kras G12D cancer treatment, aiming to address critical unmet medical needs in oncology.
Brinsupri is a first-in-class DPP1 inhibitor that has been found to inhibit activation of neutrophil enzymes that drive chronic airway inflammation in non-cystic fibrosis bronchiectasis.

Vor Bio appoints Dallan Murray as chief commercial officer, leveraging his extensive experience to drive commercialization and growth strategies for innovative therapies.

The urology MSO will become part of The Specialty Alliance.

A new CDC campaign "Free Mind" addresses rising drug overdoses and mental health issues among youth, offering vital resources for teens and caregivers.
Results from the Phase III NEPTUNUS-1 (NCT05350072) and NEPTUNUS-2 (NCT05349214) trials demonstrated that ianalumab significantly reduces disease activity and shows a favorable safety profile in adults with active Sjögren’s disease.
The journal is standing by the study, which looked into the safety of vaccines that have aluminum ingredients.
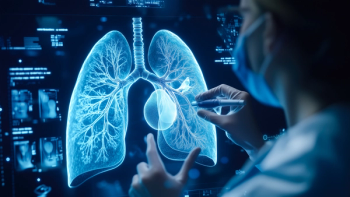
Accelerated approval was based on results from the Phase Ib Beamion-LUNG 1 trial (NCT04886804), which showed a 75% objective response rate in patients with unresectable or metastatic non-squamous non-small cell lung cancer treated with Hernexeos.

Vinay Prasad returns to lead the FDA's CBER after leaving in late July.

GLP-1 medication users must consult physicians before surgery to mitigate anesthesia risks and ensure safe pre-operative practices.

Gene Mack, CEO of Gain Therapeutics, outlines the company’s strategic approach to advancing GT-02287 through potential collaborations and financing options as they await critical Parkinson’s disease trial data.

Radella Pharmaceuticals reveals promising Phase 1b trial results for MD-18, showcasing significant weight loss and cardiometabolic health improvements.