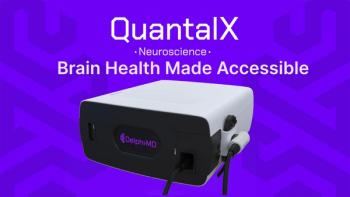
FDA’s program will provide guidance to the development of the company’s Delphi-MD technology.

FDA’s program will provide guidance to the development of the company’s Delphi-MD technology.

The technology will reportedly improve consumer engagement.
The vaccine can reportedly prevent infection from a fungal source that impacts both people and dogs.

The latest news for pharma industry insiders.
Early results have demonstrated that mRNA-3927 has led to a 70% reduction in the relative risk for metabolic decompensation events in patients with propionic acidemia.
Agreement comes as Teva aims to strengthen its biosimilar portfolio as part of its "Pivot to Growth" strategy.

The two programs will be offered to students and CVS interns.

Otsuka and Click Therapeutics co-developed Rejoyn, a digital therapeutic for MDD.

The latest news for pharma industry insiders.

Fanapt was previously approved by the FDA in 2009 as an antipsychotic treatment for schizophrenia.
Approval comes amid positive results from TRILUMINATE trial, which demonstrated improvement in 90% of participants treated for a leaky tricuspid heart valve.

A Q&A with Dr. Zeynep Ton, professor at the MIT Sloan School of Management.

The latest news for pharma industry insiders.

The two company’s will provide consumers with a platform where they can compare drug prices.
The concussion test, which shows results after 15 minutes, can help evaluate patients up to 24 hours after injury, company says.

Approval would mark first HER2-targeted therapy for biliary tract cancer in the United States.

In this Pharmaceutical Executive video interview, Optum Rx clinical pharmacist, Arash Sadeghi, discusses upcoming advancements in the drug pipeline and trends shaping the future of drug development.

The two organizations will work together to advocate for better health outcomes for children and families in North Carolina.

The latest news for pharma industry insiders.

The network will work with DiRx and MakoRX.
Treatment approved in combination with ravulizumab or eculizumab, representing minority demographic still suffering after receiving C5 inhibitor therapy for extravascular haemolysis.

In this Pharmaceutical Executive video interview, Optum Rx clinical pharmacist, Arash Sadeghi, discusses developments for non-invasive diagnostics for liver disease and how such advancements could potentially impact the landscape of conditions like NASH.

In an interview with Pharm Exec Associate Editor Don Tracy, Fady Boctor, President, Chief Commercial Officer, Petros Pharmaceuticals provides his thoughts on what the future looks like when it comes to over-the-counter (OTC) contraceptives.

The latest news for pharma industry insiders.

Company says it remains committed to advancing research for immune-mediated diseases and intends to share trial data at a later date.

The news comes less than a year after Pfizer announced that IPG would handle this role.

The study used technology developed by a variety of companies, including RYLTI.
Phase II clinical study showed that children as young as six years of age with hepatitis B virus could benefit from treatment with Vemlidy.
Deal aims to commercialize XTX301 for treating advanced solid tumors by leveraging Xilio’s tumor-activated immuno-oncology therapies.

The latest news for pharma industry insiders.