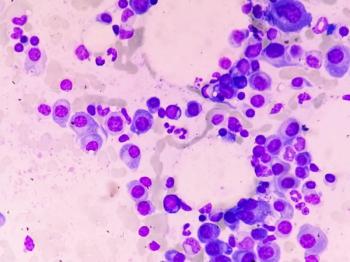
Tecvayli is an off-the-shelf bispecific antibody previously granted accelerated approval by the FDA for adults with relapsed/refractory multiple myeloma.

Tecvayli is an off-the-shelf bispecific antibody previously granted accelerated approval by the FDA for adults with relapsed/refractory multiple myeloma.
Amtagvi is the first one-time, individualized T-cell therapy approved by the FDA for any solid tumor cancer.
The FDA has assigned a Prescription Drug User Fee Act action date of August 20, 2024, for Servier’s New Drug Application for vorasidenib.
The FDA assigned the supplemental new drug application for Krazati (adagrasib) plus cetuximab in patients with locally advanced or metastatic colorectal cancer with a Prescription Drug User Fee Act goal date of June 21, 2024.
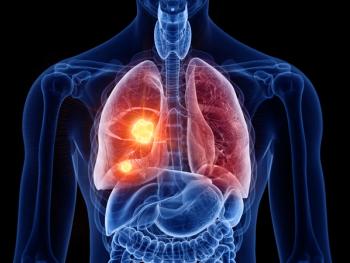
The FDA has also approved Tagrisso (osimertinib) as a monotherapy for the first-line treatment of patients with locally advanced or metastatic EGFR-mutated non-small cell lung cancer (NSCLC), locally advanced or metastatic EGFR T790M mutation-positive NSCLC, and adjuvant treatment of early-stage EGFRm NSCLC.
Amtagvi (lifileucel) becomes the first and only one-time, individualized T cell therapy to gain FDA approval for a solid tumor cancer.

Novartis’ and Roche’s Xolair (omalizumab) is indicated to treat severe allergic reactions after accidental exposure to one or more foods in individuals aged one year and above.
The FDA previously granted accelerated approval to Tepmetko for metastatic non–small cell lung cancer harboring MET exon 14 skipping alterations.

Lymphir is an immunotherapy under evaluation for the treatment of patients with relapsed/refractory cutaneous T-cell lymphoma following at least one previous line of systemic therapy.
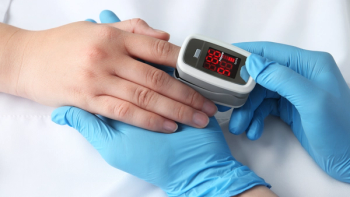
The MightySat Fingertip Pulse Oximeter is reportedly the first pulse oximeter available without a prescription.
Augtyro is a potential best-in-class tyrosine kinase inhibitor that targets ROS1- or NTRK-positive locally advanced or metastatic solid tumors.
Pancreatic adenocarcinoma is the most common form of pancreatic cancer, with more than 60,000 diagnoses per year in the United States.
BXCL701 is an oral innate immune activator being investigated in combination with Keytruda (pembrolizumab) in patients with metastatic small cell neuroendocrine prostate cancer.
In clinical trials, bepirovirsen showed the potential to address a significant unmet medical need for patients with chronic hepatitis B by reducing hepatitis B surface antigen levels and HBV DNA.
Takeda expects Eohilia single-dose stick packs will be available by the end of February for patients aged 11 years and older with eosinophilic esophagitis.

Steven Grossman, Co-founder and executive director of the Alliance for a Stronger FDA, discusses the reason for founding the alliance, its members, goals, and the resources they offer.

Data from the ENHANCE-3 trial of magrolimab in combination with azacitidine plus Venclexta showed futility and an increased risk of death in patients with acute myeloid leukemia.

Results from the Phase III CheckMate -77T trial show that Opdivo (nivolumab) plus chemotherapy followed by surgery and adjuvant Opdivo produced statistically significant and clinically meaningful improvements in event-free survival in patients with resectable stage IIA to IIIB non-small cell lung cancer.
Vepdegestrant (ARV-471) is a noval PROTAC ER degrader found to harness the body’s own natural protein disposal system to eliminate disease-causing proteins.
BioNTech SE and Duality Biologics' next-generation antibody-drug conjugate is being evaluated for patients with platinum-resistant ovarian epithelial cancer, fallopian tube cancer, or primary peritoneal cancer previously administered one to three systemic treatment regimens.
The FDA assigned a Prescription Drug User Fee Act action date of June 7, 2024, to an application that would expand the indication of Arexvy to include adults 50-59 years with an increased risk of respiratory syncytial virus-related lower respiratory tract disease.
UV1 plus ipilimumab (Yervoy) and nivolumab (Opdivo) produced a statistically significant and clinically meaningful survival improvement in patients with unresectable malignant pleural mesothelioma.

Novartis rescinds rights to to develop and commercialize multi-tyrosine kinase inhibitor dovitinib due to a material breach by Allarity for lack of financial payment.
The biologics license application for afamitresgene autoleucel, an engineered T-cell receptor drug, was assigned a PDUFA date of August 4, 2024.
Darzalex Faspro has previously been approved by the FDA for eight indications in multiple myeloma.