
Pharmaceutical Executive
To one major legal scholar, drug safety regulation isn't just about meeting standards, it's about what you take away from one group of patients in order to benefit the rest.

Pharmaceutical Executive
To one major legal scholar, drug safety regulation isn't just about meeting standards, it's about what you take away from one group of patients in order to benefit the rest.

Pharmaceutical Executive
Even when NICE has said that drugs should be available to anyone who needs them, patients still have to convince their Primary Care Trusts.

Pharmaceutical Executive
Peter Rost, former Pfizer executive turned whistleblower, isn't just at war with his old employer. He's crusading against all of pharma, an industry he likens to the mob.

Pharmaceutical Executive
Long turnaround on ad reviews adds new complications to the process of developing marketing plans.

Pharmaceutical Executive
When one company purchases another's branded trademarks as keyword search terms, the goal is to lure consumers to the buyer's site.

Pharmaceutical Executive
The Deficit Reduction Act was designed to save the government money on Medicaid. But it also has the potential to change the very way pharma companies conduct business-if they can just figure it out.

Pharmaceutical Executive
The US Department of Justice thinks that "Average Wholesale Price" should mean just what the words say-no more, no less. Why? It's never meant that before.

Pharmaceutical Executive
MSLs tend to stay put. Just three in 10 left a job after less than a year. Half held their current position for more than three years.

Pharmaceutical Executive
Scientists are worried about what they can and cannot say. Senior scientists feel that if they voice the disagreements that are important to scientific discussion, they might have to leave FDA.

Pharmaceutical Executive
In cases that reached a verdict between 2002 and 2006, the branded pharma industry won six of six cases by verdict in Delaware, but lost six of eight in New Jersey, all by summary judgement. Despite what judges say, it's no coincidence.

Pharmaceutical Executive
The world according to New Hampshire: Doctors prescribe expensive drugs because pharma reps sell them. Interfere with the selling, and you'll cut down on the prescribing. It's a plan. But for what?

Pharmaceutical Executive
Public revulsion at animal-rights extremists is damaging their cause. The majority of people deem the worst offenders to be terrorists.

Pharmaceutical Executive
Indeed, at this time, we are aware of no evidence that the practice of authorized generics has actually deterred any Paragraph IV certification or post-180 day generic entry, let alone a challenge to an invalid patent.

Pharmaceutical Executive
If I ran the FDA, I'd have a Rose Garden ceremony for all the histleblowers in my agency. No one can know where all the skeletons are buried. We ought to honor every one of those patriots.

Pharmaceutical Executive
The suit draws a comparison to the newspaper industry: Even though papers profit from disseminating information, the information in question isn't commercial.

Pharmaceutical Executive
Adaptive trials aren't just for propeller-heads anymore. They're one of the issues that need to be top-of-mind for the whole executive suite, as a driver of new processes and timelines, as a hot-spot on the budget, and as a battleground where public policy on drug safety and efficacy will be fought out.

Pharmaceutical Executive
RFID is not ready for prime time anywhere. Certainly not in the US. There is no way RFID gives you end-to-end control of the product.

Pharmaceutical Executive
California has proposed legislation for a pilot program to reinforce access to treatment for mentally ill offenders. This is a step in the right direction, but it should be the subject of national policy, not a localized effort.

Pharmaceutical Executive
Increased demand for plant-based hormones has spurred Wyeth to ask FDA to set marketing limits on compounded therapies

Pharmaceutical Executive
Home and abroad, the US Pharmacopeia is stepping up to maintain quality control. But it's not so easy. USP's Roger Williams discusses Medicare formularies, drug safety, international drug production, and the organization's changing role.
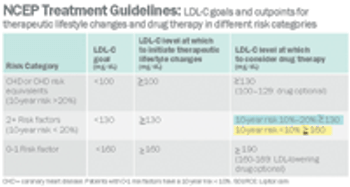
Pharmaceutical Executive
The world's largest drug manufacturer must answer off-label promotion charges brought by a new adversary. Not FDA, with its warning letters and threats of marketing sanctions, and not the Office of the Inspector General (OIG) at Health and Human Services, which often sues for fraud, forces huge settlements, and requires companies to do business under restrictive corporate integrity agreements. Instead, the company faces a class-action civil suit from insurance companies and union welfare funds, groups that, until recently, Pfizer regarded primarily as customers-or at least people who picked up the tab for customers. Now, led by the Welfare Fund of a Teamsters local from New Jersey, third-party payers are suing under RICO, the Racketeering Influenced and Corrupt Organizations Act. If their suit is successful, payers who have covered billions of dollars worth of Lipitor (atorvastatin) over the past five years will receive treble damages for the cost of off-label prescriptions. The suit may also attract..

Pharmaceutical Executive
Criminal penalties for violating the Foreign Corrupt Practices Act can be substantial. Businesses found guilty may be fined upwards of $2.5 million for each offense, or twice the amount gained as a result of the violation.

Pharmaceutical Executive
The pharmaceutical companies in the Predictive Safety Consortium have agreed to cross-test each other's laboratory methods to determine which are most effective in detecting kidney, muscle, and liver toxicity.

Pharmaceutical Executive
Where is pharma going? Toward more science, and more political pressure on science. Toward greater patient responsibility-and more regulation-by-lawsuit. And forward. Let's not forget about forward.

Pharmaceutical Executive
One health insurance company in The Netherlands is offering doctors a financial incentive to prescribe generic statins and proton pump inhibitors. Doctors and patients complained, but a court upheld the practice.