
The Big Pharma commercialization playbook doesn’t work for emerging life sciences companies bootstrapping their way up. Exploring three strategies that could be the ingredients of a winning formula for startups.


The Big Pharma commercialization playbook doesn’t work for emerging life sciences companies bootstrapping their way up. Exploring three strategies that could be the ingredients of a winning formula for startups.

Three key components to scaling quickly—while maintaining purpose.

A value deficit and potential consumer value proposition.

A Harvard Business School Healthcare Alumni Association Q&A with Martin Gonzalez, Principal of Org and Leadership Development at Google.

Omnichannel marketing can help companies improve HCP engagement to facilitate the delivery of effective treatment to patients who desperately need and deserve it.

The Octave Bioscience CEO discusses leading the company through commercialization and expansion.

Atossa Therapeutics’ CEO discusses how the new draft guidance will change the way that many women receive their breast exam results.

Conferences, congresses, and the digital discussions kicked off by those events create a halo effect that amplifies scientific information more so than other scientific activities.

Jan van de Winkel, PhD, president and CEO of Genmab, the longtime innovation engine in antibody drug development, discusses its vision for adding a new business identity: commercial driver.

Amid shifting operating dynamics in recent years, demands for external expertise have fluctuated—leaving those arrangements that mix innovation and agility likely to win out in pharma decision-making.

With the increasing adoption of COAs, the importance of standardization across training, collection, and implementation to ensure high quality and consistent data collection has become apparent in recent decades.

As the second half of 2024 begins, an industry expert discusses how mergers and acquisitions are trending in the pharma industry.

Pharmaceutical companies are seeing the benefits of being able to outsource entire functions through functional service providers.
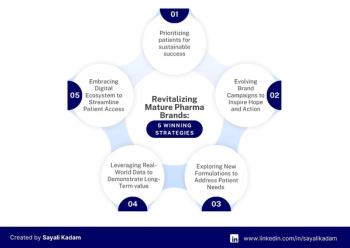
Strategies that can help maintain and grow your market share. Plus how to adapt to new competitive pressures and evolving customer needs.

Collaboration can make commercialization processes efficient and effective.

Rizzo discusses Veeva’s latest findings on how life sciences companies are utilizing this strategy to speed up uptake of new treatments.

Manufacturers must navigate complex regulatory landscapes, understand diverse cultural nuances, and prioritize patient access in each region.

Strategic imperatives for biotech firms in light of the latest market dynamics and investor behaviors.

Integrated Delivery Networks have impacted everything from drug launches to sales approaches.

As HCPs continue to face time constraints and information overload, delivering relevant and personalized content becomes increasingly critical.

Insights from Takeda, Jazz, Nestlé Health Sciences, Humana, and other healthcare executives

The global president of endomechanical and energy for Johnson & Johnson MedTech discusses the need for multidisciplinary care when treating obesity.

The CEO of Revelation Pharma discusses how compounding pharmacies may help solve the ongoing issue with drug shortages.

David Minkin, President & GM at epocrates, offers his insights into the importance of trust, first-party data, and strategic partnerships in ensuring successful marketing campaigns.

Deployments are often built on the concept that growth comes from new prescribers, which may be limiting their success.