Positive Phase III study results for Tonmya (cyclobenzaprine HCl sublingual tablets) will be basis of New Drug Application to the FDA for the management of fibromyalgia.
ViiV Announces Promising Efficacy Data for Long-Acting Cabenuva in HIV Patients With Adherence Challenges to Daily ART

Teva Reports Positive Data from Anti-TL1A Antibody for Inflammatory Bowel Diseases

Positive Phase III study results for Tonmya (cyclobenzaprine HCl sublingual tablets) will be basis of New Drug Application to the FDA for the management of fibromyalgia.

Looking to hold an advisory board? Here are a few suggestions.
As therapeutics become more personalized and targeted, technology, such as open-source software, can help companies innovate.
BXCL701 is an oral innate immune activator being investigated in combination with Keytruda (pembrolizumab) in patients with metastatic small cell neuroendocrine prostate cancer.
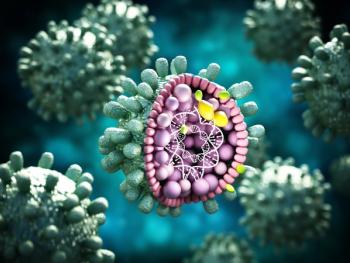
In clinical trials, bepirovirsen showed the potential to address a significant unmet medical need for patients with chronic hepatitis B by reducing hepatitis B surface antigen levels and HBV DNA.

Novel oral orexin receptor 2 agonist produced statistically significant and clinically meaningful improvements in wakefulness compared with placebo in patients with narcolepsy type 1.

Researchers evaluate a potential association between the use of phosphodiesterase type 5 inhibitors for erectile dysfunction and a reduced risk of Alzheimer disease.
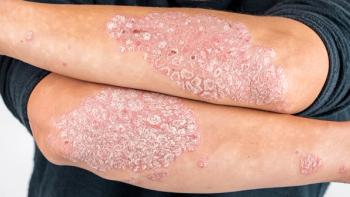
JNJ-2113, an oral IL-23–receptor antagonist peptide, showed consistency across clinician and patient-reported outcomes in patients with moderate-to-severe plaque psoriasis.

Data from the ENHANCE-3 trial of magrolimab in combination with azacitidine plus Venclexta showed futility and an increased risk of death in patients with acute myeloid leukemia.
The biopharma company’s Phase 3 trial for its acoramidis product generated statistically significant results.

The Maryland facility will create cell therapies for use in future cancer trials.

Results from the Phase III CheckMate -77T trial show that Opdivo (nivolumab) plus chemotherapy followed by surgery and adjuvant Opdivo produced statistically significant and clinically meaningful improvements in event-free survival in patients with resectable stage IIA to IIIB non-small cell lung cancer.
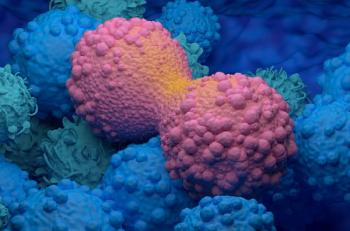
Vepdegestrant (ARV-471) is a noval PROTAC ER degrader found to harness the body’s own natural protein disposal system to eliminate disease-causing proteins.

In a Phase I trial, data suggest that Amgen's maridebart cafraglutide (MariTide) may allow patients to take lower and less frequent doses over time while still maintaining significant weight loss.
Patients with relapsed or refractory multiple myeloma administered Blenrep combined with bortezomib plus dexamethasone experienced a 59% reduction in the risk of disease progression or death compared with the standard of care.
BioNTech SE and Duality Biologics' next-generation antibody-drug conjugate is being evaluated for patients with platinum-resistant ovarian epithelial cancer, fallopian tube cancer, or primary peritoneal cancer previously administered one to three systemic treatment regimens.
The FDA assigned a Prescription Drug User Fee Act action date of June 7, 2024, to an application that would expand the indication of Arexvy to include adults 50-59 years with an increased risk of respiratory syncytial virus-related lower respiratory tract disease.
UV1 plus ipilimumab (Yervoy) and nivolumab (Opdivo) produced a statistically significant and clinically meaningful survival improvement in patients with unresectable malignant pleural mesothelioma.

The era of big pharma as product-first companies must end, as services become the larger priority.
The biologics license application for afamitresgene autoleucel, an engineered T-cell receptor drug, was assigned a PDUFA date of August 4, 2024.
BST02 is the first tumor-infiltrating lymphocyte therapy for the treatment of all types of liver cancer to have advanced to the clinical trial stage.

DELFI-Tumor Fraction assay was developed to improve noninvasive assessment of tumor burden and monitoring of treatment efficacy and resistance in patients with advanced cancers.
Rusfertide is currently in a pivotal Phase III clinical trial as a potential first-in-class treatment for polycythemia vera.
Vabysmo is the first bispecific antibody approved to treat ocular conditions such as diabetic macular edema and wet age-related macular degeneration.

Including patient and family feedback in our discussions will help shape our understanding of their perception of provided care and services and how we operate as a whole.