R&D/Clinical Trials
Latest News
VolitonRx’s Cancer Test Could be Used to Detect 21 Different Cancers, Study Suggests
Latest Videos

More News

Company executives provide updates on cancer treatment pelareorep and other elements of the company’s pipeline.
In the Phase III CALYPSO trial, eneboparatide demonstrated statistical significance in achieving albumin-adjusted serum calcium normalization while eliminating the need for active vitamin D and oral calcium therapy in chronic hypoparathyroidism.

Jonathon Whitton, AuD, PhD, VP, auditory global program head, Regeneron, discusses how the focus of technology development is shifting toward ensuring safety and continuous hearing for children.
Data from the Phase III MINT trial found that Uplizna demonstrated a greater reduction in Myasthenia Gravis Activities of Daily Living score compared to placebo at week 26.

Artiva Biotherapeutics' CEO Fred Aslan, MD, explains the strategic advantages of pursuing both a company-sponsored trial and an investigator-initiated trial simultaneously.
New real-world and implementation study data highlight the efficacy of ViiV’s long-acting injectables for HIV prevention and treatment, with Apretude showing zero HIV acquisitions and Cabenuva maintaining high viral suppression rates.

Immunotherapy pioneer Mark Frohlich, MD, CEO of Indapta Therapeutics, discusses the promise of harnessing natural killer cells to reshape treatment for cancer and autoimmune disease.

Artiva Biotherapeutics’ CEO Fred Aslan, MD, identifies data points the company is prioritizing to demonstrate that results from certain trials will translate to similar efficacy across a range of autoimmune conditions.
Topline results from the Phase III VERITAC-2 trial found that vepdegestrant provided a statistically significant and clinically meaningful improvement in progression-free survival in patients with ER+/HER2- advanced or metastatic breast cancer.

Dr. Dina Radenkovic, CEO of Gameto, discusses the use of iPSCs in fertility treatments, the most significant scientific hurdles they've encountered in developing and scaling these therapies (particularly in relation to ovarian aging), and how they're addressing them.
Results from multiple Phase III trials of Icotrokinra in moderate-to-severe plaque psoriasis and a Phase IIb trial in ulcerative colitis successfully met all primary endpoints.

What biopharma companies need to know about this evolving regulatory pathway.

Aliya Omer, VP, US franchise head, breast cancer, AstraZeneca, discusses Lynparza’s role in reducing the risk of invasive disease recurrence or death in HR-positive breast cancer patients.

Artiva Biotherapeutics' CEO Fred Aslan, MD, discusses the concept of an immune reset and what the company is tracking in trials to assess its potential.

Jonathon Whitton, AuD, PhD, VP, auditory global program head, Regeneron, discusses how key regulatory designations, such as the FDA's Regenerative Medicine Advanced Therapy designation, facilitate accelerated development and commercialization of DB-OTO through increased collaboration with regulators.

Artiva Biotherapeutics' CEO Fred Aslan, MD, discusses two ongoing trials for autoimmunity indications in the US and how AlloNK differs from traditional B-cell depletion strategies.
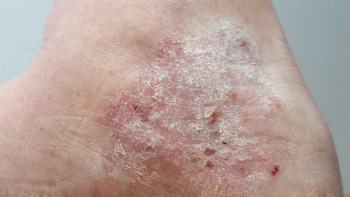
Five-year data from Phase III trials demonstrated that 67.7% of patients moderate-to-severe plaque psoriasis treated with UCB’s Bimzelx achieved complete skin clearance.

Clinical trial results found that a majority of patients with atopic dermatitis who were treated with Ebgylss achieved complete or near complete skin clearance at three years on a single monthly maintenance dose.

Jonathon Whitton, AuD, PhD, VP, auditory global program head, Regeneron, discusses the promising results of DB-OTO in the CHORD trial for children with otoferlin-related hearing loss.
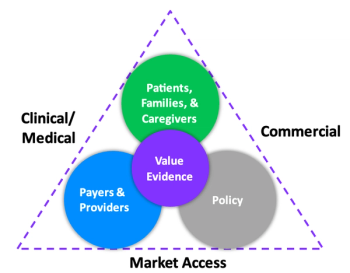
The recent shift in Health Economics and Outcomes Research functions across major pharmaceutical companies highlights a lack of understanding of its value.

Dr. Hernan Bazan, CEO of South Rampart Pharma, discusses his company’s current pipeline.

Aliya Omer, VP, US franchise head, breast cancer, AstraZeneca, discusses how results of the trial compare with other treatments for gBRCAm HER2-negative high-risk early breast cancer.

Under terms of the deal, Gubra will receive an initial $350 million payment and could earn up to $1.875 billion more in development and sales milestones for the potential weight loss drug.
FP008 is designed for patients with solid tumors that have not responded to existing immunotherapies targeting PD-1.

The Alpha Cognition executive discusses recent developments that positively impact the upcoming launch for ZUNVEYL.